Advertisements
Advertisements
Question
Give the structure of the product you would expect when the following alcohol reacts with SOCl2.
2-Methylbutan-2-ol
Advertisements
Solution
\[\begin{array}{cc}
\ce{CH3}\phantom{.............................................}\ce{CH3}\phantom{........................}\\
|\phantom{...................................................}|\phantom{............................}\\
\ce{CH3 - C - CH2CH3 + SOCl2 ->[\Delta]CH3 - C - CH2CH3 + SO2 + HCl}\\
|\phantom{...................................................}|\phantom{............................}\\
\ce{OH}\phantom{...................................}\ce{\underset{2-chloro-2-methylbutane}{Cl}}\phantom{................}\\
\end{array}\]
APPEARS IN
RELATED QUESTIONS
Write the equation involved in the acetylation of Salicylic acid.
Give the structure of the product you would expect when the following alcohol reacts with HCl–ZnCl2.
Butan-1-ol
Explain how does the −OH group attached to a carbon of benzene ring activate it towards electrophilic substitution?
Acidity of phenol is due to ____________.
In the following compounds:
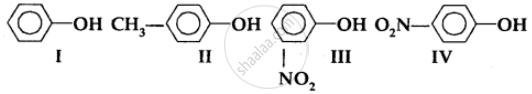
The order of acidity is
In CH3CH2OH, the bond that undergoes heterolytical change most readily is ____________.
Strength of acidity is in order:

Which of the following statements is true:
Out of o-nitrophenol and o-cresol which is more acidic?
Assertion: o-Nitrophenol is less soluble in water than the m- and p-isomers.
Reason: m- and p- Nitrophenols exist as associated molecules.
Phenol is used in the manufacture of
Arrange the following in the increasing order of their property indicated:
4-Nitrobenzoic acid, benzoic acid, 3,4-Dinitrobenzoic acid, 4-Methoxy benzoic acid (Acid strength)
For the pair phenol and cyclohexanol, answer the following:
Why is phenol more acidic than cyclohexanol?
Give the structure of the product you would expect when the following alcohol reacts with HBr.
2-Methylbutan-2-ol
Compare acidity of phenol with that of ethanol.
