Advertisements
Advertisements
Question
Write the equation involved in the acetylation of Salicylic acid.
Advertisements
Solution
Acetylation of Salicylic acid:
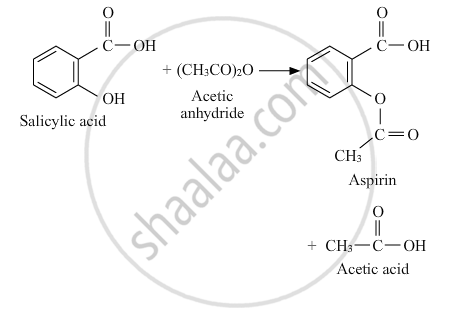
APPEARS IN
RELATED QUESTIONS
Give two reactions that show the acidic nature of phenol.
Account for the following:
o-nitrophenol is more steam volatile than p-nitrophenol.
Intermolecular hydrogen bonding is strongest in ______.
Phenols do not react with one of the following:
Phenol is more acidic than alcohol because ____________.
Which of the following compounds is most acidic?
Out of o-nitrophenol and o-cresol which is more acidic?
Give the structure of the product you would expect when the following alcohol reacts with HBr.
Butan-1-ol
Give the structure of the product you would expect when the following alcohol reacts with SOCl2.
Butan-1-ol
Compare acidity of phenol with that of ethanol.
