Advertisements
Advertisements
Question
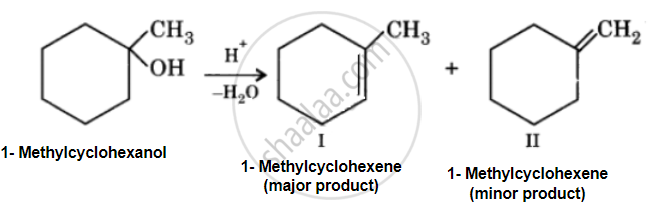
Predict the major product of acid catalysed dehydration of 1-methylcyclohexanol.
Advertisements
Solution
Acid catalyzed dehydration of 1-methylcyclohexanol can give two products, I and II. Since product (I) is more highly substituted, it is the major product according to the Setzeff rule.
APPEARS IN
RELATED QUESTIONS
Name the reagents used in the following reactions:

Name the reagents used in the following reactions:

Show how will you synthesize 1-phenylethanol from a suitable alkene.
How is the following conversion carried out?
\[\ce{Propene -> Propan-2-ol}\]
How is the following conversion carried out?
\[\ce{Methyl magnesium bromide → 2-Methylpropan-2-ol}\]
Show how you would synthesise the following alcohol from an appropriate alkene?
How will you convert: Phenol to 2, 4, 6 − trinitrophenol?
Aldehydes are reduced to the corresponding alcohols by the addition of hydrogen in the presence of catalysts to form ____________.
Benzaldehyde differs from acetaldehyde in that:
Most readily hydrolysed halide is:-
The best reagent to convert pent 3 – en 2 – 01 into pent 3 – in – 2 – one is
Glycerol as a trimester present in
The best reagent to convert pent - 3 - en - 2 - ol into pent - 3 - en - 2 one is ______.
An aldehyde isomeric with allyl alcohol gives phenyl hydrazone. Pick out a ketone that too gives a phenyl hydrazone containing the same percentage of nitrogen.
\[\ce{? ->[\Delta, CN-][EtOH, H2O]}\] Benzoin.
The reactant is obtained by dry distillation of the calcium salts of the following pairs:
Given below are two statements:
Statement I: On heating with KHSO4, glycerol is dehydrated and acrolein is formed.
Statement II: Acrolein has a fruity odour and can be used to test glycerol's presence.
Choose the correct option.
Ceric aminonium nitrate and CHCl3/alc. KOH are used for the identification of functional groups present in ______ and ______ respectively.
Write the mechanism of acid dehydration of ethanol to yield ethene.
