Advertisements
Advertisements
प्रश्न
Predict the major product of acid catalysed dehydration of 1-methylcyclohexanol.
Advertisements
उत्तर १
Acid catalyzed dehydration of 1-methylcyclohexanol can give two products, I and II. Since product (I) is more highly substituted, it is the major product according to the Setzeff rule.
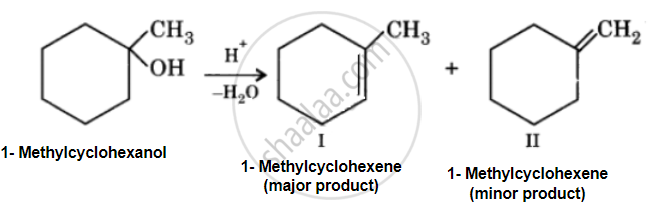
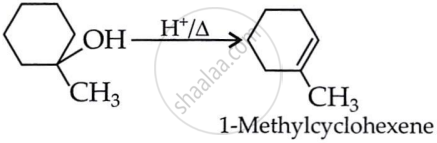
उत्तर २
APPEARS IN
संबंधित प्रश्न
How will you convert: Propene to Propan-2-ol?
Write the main product(s) in each of the following reactions:

How do you convert the following : Propan-2-ol to 2-methylpropan-2-ol
Write the structures of the main products when acetone (CH3 − CO − CH3) reacts with the following reagents :
CH3MgBr and then H3O+
Show how is the following alcohol prepared by the reaction of a suitable Grignard reagent on methanal?
\[\begin{array}{cc}
\ce{CH3 - CH - CH2OH}\\
|\phantom{........}\\
\ce{CH3}\phantom{....}\\
\end{array}\]
Name the reagent used in the following reaction:
Dehydration of propan-2-ol to propene.
Write the structures of A, B and C in the following reactions :
Alkenes convert into alcohols by ____________.
Commercially carboxylic acids are reduced to alcohols by converting them to the ______.
Acetone reacts with Grignard reagent to form:
Magnesium wire continues to burn in the atmosphere of CO2 because
The reagent used for dehydration of an alcohol is
When alcohol react with concentrated H2SO4 intermediate compound formed is
Glycerol is used in the manufacture
To synthesise 1.0 mole of 2-methylpropan-2-ol from Ethylethanoate ______ equivalents of CH3MgBr reagent will be required. (Integer value)
An aldehyde isomeric with allyl alcohol gives phenyl hydrazone. Pick out a ketone that too gives a phenyl hydrazone containing the same percentage of nitrogen.
\[\ce{C3H8O ->[{[O]}][K2Cr2O7/H2SO4] C3H6O ->[I2 + NaOH(aq.)] CHI3}\]
In this reaction the first compound is:
The major product of the following reaction is:
\[\begin{array}{cc}
\ce{Cl}\phantom{.........................}\\
|\phantom{..........................}\\
\ce{CH3 - CH - CH3 ->[(i) Alc. KOH][(ii) HBr/peroxide (iii) aq. KOH]}
\end{array}\]
Write the mechanism of acid dehydration of ethanol to yield ethene.
