Advertisements
Advertisements
प्रश्न
Name the reagent used in the following reaction:
Dehydration of propan-2-ol to propene.
Advertisements
उत्तर
Conc. H2SO4 or H3PO4 at 433-443 K is used as a reagent in the dehydration of propan-2-ol to propene.
APPEARS IN
संबंधित प्रश्न
Predict the major product of acid catalysed dehydration of butan-1-ol.
What is meant by hydroboration-oxidation reaction? Illustrate it with an example.
Show how will you synthesize 1-phenylethanol from a suitable alkene.
How is the following conversion carried out?
\[\ce{Benzyl chloride -> Benzyl alcohol}\]
Name the reagent used in the following reaction:
Butan-2-one to butan-2-ol.
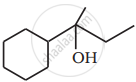
Show how you would synthesise the following alcohol from an appropriate alkene?
Write the structure of main compounds A and B in the following reaction:
\[\ce{CH3CH2CN->[CH3MgBRH/3O+]A->[LiAIH4]B}\]
Aldehydes are reduced to the corresponding alcohols by the addition of hydrogen in the presence of catalysts to form ____________.
How can propan-2-one be converted into tert- butyl alcohol?
Why is the reactivity of all the three classes of alcohols with conc. \[\ce{HCl}\] and \[\ce{ZnCl2}\] (Lucas reagent) different?
Most readily hydrolysed halide is:-
When alcohol react with concentrated H2SO4 intermediate compound formed is
When glycol is heated with dicorboxylic acid the product are
The best reagent to convert pent - 3 - en - 2 - ol into pent - 3 - en - 2 one is ______.
The major product of acid catalysed dehydration of 1-methylcyclohexanol is ______.
To synthesise 1.0 mole of 2-methylpropan-2-ol from Ethylethanoate ______ equivalents of CH3MgBr reagent will be required. (Integer value)
Given below are two statements:
Statement I: On heating with KHSO4, glycerol is dehydrated and acrolein is formed.
Statement II: Acrolein has a fruity odour and can be used to test glycerol's presence.
Choose the correct option.
Ceric aminonium nitrate and CHCl3/alc. KOH are used for the identification of functional groups present in ______ and ______ respectively.
Write the mechanism of acid dehydration of ethanol to yield ethene.
How are the following conversions carried out?
\[\ce{Methyl magnesium bromide ->2-Methylpropan-2-ol}\]
