Advertisements
Advertisements
प्रश्न
Name the reagent used in the following reaction:
Butan-2-one to butan-2-ol.
Advertisements
उत्तर
H2/Ni or NaBH4 or LiAIH4 is used as a reagent in butan-2-one to butan-2-ol.
APPEARS IN
संबंधित प्रश्न
Name the reagents used in the following reactions:

Name the reagents used in the following reactions:

Write the structures of the main products when acetone (CH3 − CO − CH3) reacts with the following reagents :
CH3MgBr and then H3O+
Show how is the following alcohol prepared by the reaction of a suitable Grignard reagent on methanal?

Predict the major product of acid catalysed dehydration of butan-1-ol.
What is meant by hydroboration-oxidation reaction? Illustrate it with an example.
Show how will you synthesize 1-phenylethanol from a suitable alkene.
How is the following conversion carried out?
\[\ce{Ethyl magnesium chloride -> Propan-1-ol}\]

Show how you would synthesise the following alcohol from an appropriate alkene?

Show how you would synthesise the following alcohol from an appropriate alkene?
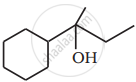
Name the reagents used in the following reactions:

Ketones react with Grignard reagent to produce ____________.
Explain a process in which a biocatalyst is used in industrial preparation of a compound known to you.
Select the acid(s) which cannot be prepared by Grignard reagent.
Glycerol is used in the manufacture
The best reagent to convert pent - 3 - en - 2 - ol into pent - 3 - en - 2 one is ______.


The products "A" and "B" formed in above reactions are:
\[\ce{? ->[\Delta, CN-][EtOH, H2O]}\] Benzoin.
The reactant is obtained by dry distillation of the calcium salts of the following pairs:
