Advertisements
Advertisements
प्रश्न
Show how you would synthesise the following alcohol from an appropriate alkene?
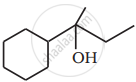
Advertisements
उत्तर
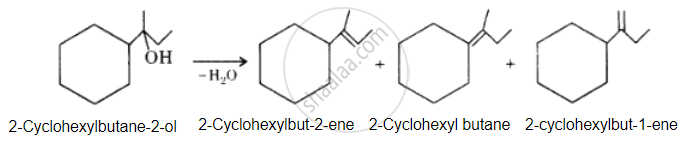
The desired alcohol is obtained from any of these alkenes by adding H2O in the presence of acid.
APPEARS IN
संबंधित प्रश्न
Show how is the following alcohol prepared by the reaction of a suitable Grignard reagent on methanal?

Predict the major product of acid catalysed dehydration of 1-methylcyclohexanol.
What is meant by hydroboration-oxidation reaction? Illustrate it with an example.
Show how will you synthesize 1-phenylethanol from a suitable alkene.
How is the following conversion carried out?
\[\ce{Methyl magnesium bromide → 2-Methylpropan-2-ol}\]

Name the reagents used in the following reactions:

Write the structure of main compounds A and B in the following reaction:
\[\ce{CH3CH2CN->[CH3MgBRH/3O+]A->[LiAIH4]B}\]
How can propan-2-one be converted into tert- butyl alcohol?
The Wittig reaction is a reaction between a carbonyl compound (aldehyde or ketone only) and a species known as a phosphoniumylide. What is the expected final product in the Wittig reaction?
Magnesium wire continues to burn in the atmosphere of CO2 because
The reagent used for dehydration of an alcohol is
Most readily hydrolysed halide is:-
Glycerol is used in the manufacture
When glycol is heated with dicorboxylic acid the product are
The major product of acid catalysed dehydration of 1-methylcyclohexanol is ______.
\[\ce{C3H8O ->[{[O]}][K2Cr2O7/H2SO4] C3H6O ->[I2 + NaOH(aq.)] CHI3}\]
In this reaction the first compound is:
For distinction between \[\ce{CH3CHO}\] and \[\ce{C6H5CHO}\] the reagent used is ______.
How are the following conversions carried out?
\[\ce{Methyl magnesium bromide ->2-Methylpropan-2-ol}\]
How are the following conversions carried out?
\[\ce{Methyl magnesium bromide->2-Methylpropan-2-ol}\]
