Advertisements
Advertisements
प्रश्न
Show how you would synthesise the following alcohol from an appropriate alkene?
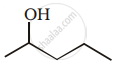
Advertisements
उत्तर
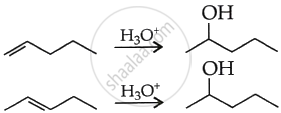
APPEARS IN
संबंधित प्रश्न
Name the reagents used in the following reactions:

Write the structures of the main products when acetone (CH3 − CO − CH3) reacts with the following reagents :
CH3MgBr and then H3O+
Predict the major product of acid catalysed dehydration of 1-methylcyclohexanol.
Predict the major product of acid catalysed dehydration of butan-1-ol.
How is the following conversion carried out?
\[\ce{Ethyl magnesium chloride -> Propan-1-ol}\]
Name the reagent used in the following reaction:
Butan-2-one to butan-2-ol.
Show how you would synthesise the following alcohol from an appropriate alkene?
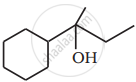
Name the reagents used in the following reactions:

Write the structures of A, B and C in the following reactions :
Ketones react with Grignard reagent to produce ____________.
Benzaldehyde differs from acetaldehyde in that:
Why is the reactivity of all the three classes of alcohols with conc. \[\ce{HCl}\] and \[\ce{ZnCl2}\] (Lucas reagent) different?
Most readily hydrolysed halide is:-
The best reagent to convert pent 3 – en 2 – 01 into pent 3 – in – 2 – one is
Alkaline hydrolysis of an alkyl halide can be preferably carried out using ______.


The products "A" and "B" formed in above reactions are:
How are the following conversions carried out?
\[\ce{Methyl magnesium bromide ->2-Methylpropan-2-ol}\]
