Advertisements
Advertisements
प्रश्न
Show how you would synthesise the following alcohol from an appropriate alkene?

Advertisements
उत्तर
The given alcohols can be synthesised by applying Markovnikov’s rule of acid-catalysed hydration of appropriate alkenes.
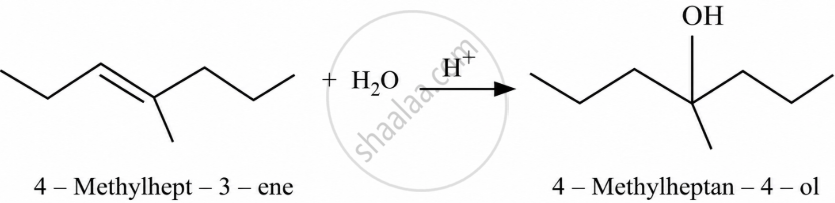
APPEARS IN
संबंधित प्रश्न
Name the reagents used in the following reactions:

Name the reagents used in the following reactions:

How do you convert the following : Propan-2-ol to 2-methylpropan-2-ol
Show how is the following alcohol prepared by the reaction of a suitable Grignard reagent on methanal?

Predict the major product of acid catalysed dehydration of 1-methylcyclohexanol.
Predict the major product of acid catalysed dehydration of butan-1-ol.
Show how will you synthesize 1-phenylethanol from a suitable alkene.
How is the following conversion carried out?
\[\ce{Propene -> Propan-2-ol}\]
How is the following conversion carried out?
\[\ce{Ethyl magnesium chloride -> Propan-1-ol}\]
Name the reagent used in the following reaction:
Butan-2-one to butan-2-ol.
Primary alcohols are prepared by the reduction of carboxylic acids. Though lithium aluminium hydride is a strong reducing agent, it is not used in the reaction. This is so because:
Monochlorination of toluene in sunlight followed by hydrolysis by aq. \[\ce{NaOH}\] yields.
Acetone reacts with Grignard reagent to form:
Glycerol is used in the manufacture
To synthesise 1.0 mole of 2-methylpropan-2-ol from Ethylethanoate ______ equivalents of CH3MgBr reagent will be required. (Integer value)


The products "A" and "B" formed in above reactions are:
An aldehyde isomeric with allyl alcohol gives phenyl hydrazone. Pick out a ketone that too gives a phenyl hydrazone containing the same percentage of nitrogen.
How are the following conversions carried out?
\[\ce{Methyl magnesium bromide ->2-Methylpropan-2-ol}\]
How are the following conversions carried out?
\[\ce{Methyl magnesium bromide->2-Methylpropan-2-ol}\]
