Advertisements
Advertisements
प्रश्न

Advertisements
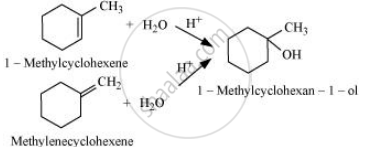
उत्तर
The given alcohols can be synthesized by applying Markovnikov’s rule of acid-catalyzed hydration of appropriate alkenes.
APPEARS IN
संबंधित प्रश्न
Predict the major product of acid catalysed dehydration of butan-1-ol.
Write the mechanism of hydration of ethene to yield ethanol.
Show how you would synthesise the following alcohol from an appropriate alkene?

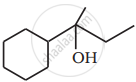
Show how you would synthesise the following alcohol from an appropriate alkene?
How will you convert: Phenol to 2, 4, 6 − trinitrophenol?
Ketones react with Grignard reagent to produce ____________.
Primary alcohols are prepared by the reduction of carboxylic acids. Though lithium aluminium hydride is a strong reducing agent, it is not used in the reaction. This is so because:
Ethyl alcohol can be prepared from Grignard reagent by the reaction of ____________.
Alkenes convert into alcohols by ____________.
Commercially carboxylic acids are reduced to alcohols by converting them to the ______.
Acetone reacts with Grignard reagent to form:
Identify ‘C’ in the following:

Magnesium wire continues to burn in the atmosphere of CO2 because
The reagent used for dehydration of an alcohol is
The best reagent to convert pent 3 – en 2 – 01 into pent 3 – in – 2 – one is
Glycerol is used in the manufacture
Glycerol as a trimester present in
When glycol is heated with dicorboxylic acid the product are
How are the following conversion carried out?
\[\ce{Methyl magnesium bromide -> 2-Methylpropan-2-ol}\]
