Advertisements
Advertisements
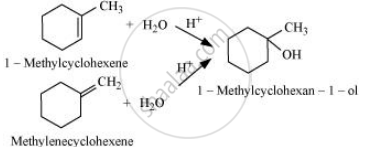
Question

Advertisements
Solution
The given alcohols can be synthesized by applying Markovnikov’s rule of acid-catalyzed hydration of appropriate alkenes.
APPEARS IN
RELATED QUESTIONS
Write the main product(s) in each of the following reactions:

How do you convert the following : Propan-2-ol to 2-methylpropan-2-ol
Show how is the following alcohol prepared by the reaction of a suitable Grignard reagent on methanal?
\[\begin{array}{cc}
\ce{CH3 - CH - CH2OH}\\
|\phantom{......}\\
\ce{CH3}\phantom{...}
\end{array}\]
Predict the major product of acid catalysed dehydration of 1-methylcyclohexanol.
How is the following conversion carried out?
\[\ce{Propene -> Propan-2-ol}\]
How is the following conversion carried out?
\[\ce{Benzyl chloride -> Benzyl alcohol}\]
How is the following conversion carried out?
\[\ce{Ethyl magnesium chloride -> Propan-1-ol}\]
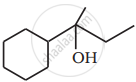
Show how you would synthesise the following alcohol from an appropriate alkene?

Show how you would synthesise the following alcohol from an appropriate alkene?
Name the reagents used in the following reactions:

Monochlorination of toluene in sunlight followed by hydrolysis by aq. \[\ce{NaOH}\] yields.
Alkenes convert into alcohols by ____________.
Acetone reacts with Grignard reagent to form:
How can propan-2-one be converted into tert- butyl alcohol?
The Wittig reaction is a reaction between a carbonyl compound (aldehyde or ketone only) and a species known as a phosphoniumylide. What is the expected final product in the Wittig reaction?
The reagent used for dehydration of an alcohol is
Most readily hydrolysed halide is:-
The major product of the following reaction is:
\[\begin{array}{cc}
\ce{Cl}\phantom{.........................}\\
|\phantom{..........................}\\
\ce{CH3 - CH - CH3 ->[(i) Alc. KOH][(ii) HBr/peroxide (iii) aq. KOH]}
\end{array}\]
Ceric aminonium nitrate and CHCl3/alc. KOH are used for the identification of functional groups present in ______ and ______ respectively.
Write the mechanism of acid dehydration of ethanol to yield ethene.
