Advertisements
Advertisements
Question
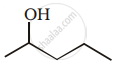
Show how you would synthesise the following alcohol from an appropriate alkene?
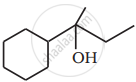
Advertisements
Solution
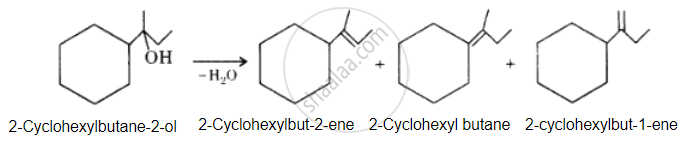
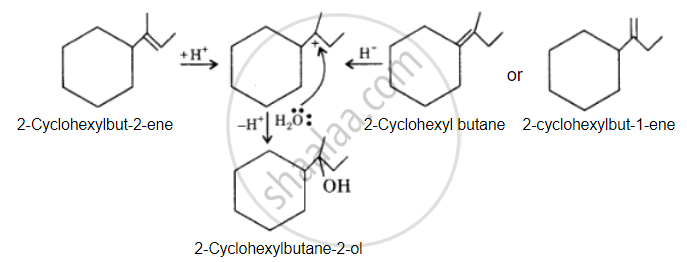
The desired alcohol is obtained from any of these alkenes by adding H2O in the presence of acid.
APPEARS IN
RELATED QUESTIONS
Name the reagents used in the following reactions:

Write the main product(s) in each of the following reactions:

Predict the major product of acid catalysed dehydration of 1-methylcyclohexanol.
What is meant by hydroboration-oxidation reaction? Illustrate it with an example.
How is the following conversion carried out?
\[\ce{Ethyl magnesium chloride -> Propan-1-ol}\]

Show how you would synthesise the following alcohol from an appropriate alkene?

Show how you would synthesise the following alcohol from an appropriate alkene?
Write the structure of main compounds A and B in the following reaction:
\[\ce{CH3CH2CN->[CH3MgBRH/3O+]A->[LiAIH4]B}\]
Primary alcohols are prepared by the reduction of carboxylic acids. Though lithium aluminium hydride is a strong reducing agent, it is not used in the reaction. This is so because:
Benzaldehyde differs from acetaldehyde in that:
Ethyl alcohol can be prepared from Grignard reagent by the reaction of ____________.
Which of the following reacts with NaOH to give alcohol?
Explain a process in which a biocatalyst is used in industrial preparation of a compound known to you.
Carboxylic acids are more acidic than phenol and alcohol because of
A compound does not react with 2, 4 dinitrophenylhydrazine, the compound is:
\[\ce{? ->[\Delta, CN-][EtOH, H2O]}\] Benzoin.
The reactant is obtained by dry distillation of the calcium salts of the following pairs:
The major product of the following reaction is:
\[\begin{array}{cc}
\ce{Cl}\phantom{.........................}\\
|\phantom{..........................}\\
\ce{CH3 - CH - CH3 ->[(i) Alc. KOH][(ii) HBr/peroxide (iii) aq. KOH]}
\end{array}\]
How are the following conversions carried out?
\[\ce{Methyl magnesium bromide ->2-Methylpropan-2-ol}\]
