Advertisements
Advertisements
प्रश्न
Show how you would synthesise the following alcohol from an appropriate alkene?
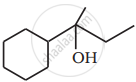
Advertisements
उत्तर
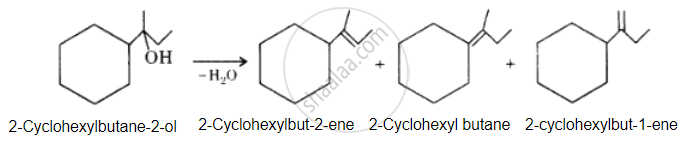
The desired alcohol is obtained from any of these alkenes by adding H2O in the presence of acid.
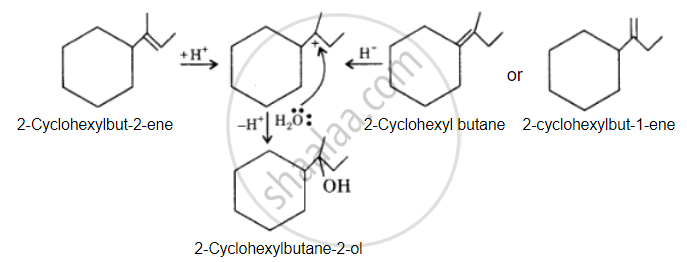
APPEARS IN
संबंधित प्रश्न
How will you convert: Propene to Propan-2-ol?
Name the reagents used in the following reactions:

Predict the major product of acid catalysed dehydration of butan-1-ol.
How is the following conversion carried out?
\[\ce{Benzyl chloride -> Benzyl alcohol}\]
How is the following conversion carried out?
\[\ce{Ethyl magnesium chloride -> Propan-1-ol}\]
How will you convert: Phenol to 2, 4, 6 − trinitrophenol?
Aldehydes react with Grignard reagent to produce ____________.
Ethyl alcohol can be prepared from Grignard reagent by the reaction of ____________.
Explain a process in which a biocatalyst is used in industrial preparation of a compound known to you.
Select the acid(s) which cannot be prepared by Grignard reagent.
Carboxylic acids are more acidic than phenol and alcohol because of
A compound does not react with 2, 4 dinitrophenylhydrazine, the compound is:
Most readily hydrolysed halide is:-
Alkaline hydrolysis of an alkyl halide can be preferably carried out using ______.
An aldehyde isomeric with allyl alcohol gives phenyl hydrazone. Pick out a ketone that too gives a phenyl hydrazone containing the same percentage of nitrogen.
The major product of the following reaction is:
\[\begin{array}{cc}
\ce{Cl}\phantom{.........................}\\
|\phantom{..........................}\\
\ce{CH3 - CH - CH3 ->[(i) Alc. KOH][(ii) HBr/peroxide (iii) aq. KOH]}
\end{array}\]
How are the following conversions carried out?
\[\ce{Methyl magnesium bromide->2-Methylpropan-2-ol}\]
