Advertisements
Advertisements
Question
How is the following conversion carried out?
\[\ce{Ethyl magnesium chloride -> Propan-1-ol}\]
Advertisements
Solution 1
When ethyl magnesium chloride is treated with methanal, an adduct produces propan-1-ol on hydrolysis.
\[\begin{array}{cc}
\ce{H}\phantom{.................................}\\
\backslash\phantom{..............................}\\
\ce{C = O + C2H5 - MgCl ->}\phantom{.}\\
/\phantom{..............................}\\
\ce{H}\phantom{.................................}
\end{array}\]\[\begin{bmatrix}
\ce{CH2 - \overset{-}{O}\overset{+}{M}gCl}\\
|\phantom{.............}\\
\ce{C2H5}\phantom{.........}\\
\ce{Adduct}
\end{bmatrix}\]\[\begin{array}{cc}
\ce{->[H2O]}
\ce{\underset{Propan-1-ol}{C3H7 - OH} + Mg(OH)Cl}
\end{array}\]
Solution 2
\[\begin{array}{cc}
\ce{O}\phantom{..........................................................................}\\
||\phantom{..........................................................................}\\
\ce{\underset{chloride}{\underset{Ethyl magnesium}{H3C - CH2 - MgCl}} + C ->[Dry ether] H3C - CH2 - CH2 - OMgCl ->[H3O+] \underset{Propanol}{CH3 - CH2 - CH2 - OH}\\
/\phantom{.}\backslash}\phantom{..........................................................................}\\
\ce{\underset{Formaldehyde}{\ce{H}\phantom{....}\ce{H}}}\phantom{..........................................................................}\\
\end{array}\]
APPEARS IN
RELATED QUESTIONS
How will you convert: Propene to Propan-2-ol?
Name the reagents used in the following reactions:

How do you convert the following : Propan-2-ol to 2-methylpropan-2-ol
Write the structures of the main products when acetone (CH3 − CO − CH3) reacts with the following reagents :
CH3MgBr and then H3O+
Predict the major product of acid catalysed dehydration of 1-methylcyclohexanol.
How is the following conversion carried out?
\[\ce{Propene -> Propan-2-ol}\]
Show how you would synthesise the following alcohol from an appropriate alkene?
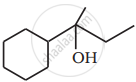
Primary alcohols are prepared by the reduction of carboxylic acids. Though lithium aluminium hydride is a strong reducing agent, it is not used in the reaction. This is so because:
Which of the following reacts with NaOH to give alcohol?
Acetone reacts with Grignard reagent to form:
Identify ‘C’ in the following:

How can propan-2-one be converted into tert- butyl alcohol?
Why is the reactivity of all the three classes of alcohols with conc. \[\ce{HCl}\] and \[\ce{ZnCl2}\] (Lucas reagent) different?
The Wittig reaction is a reaction between a carbonyl compound (aldehyde or ketone only) and a species known as a phosphoniumylide. What is the expected final product in the Wittig reaction?
When glycol is heated with dicorboxylic acid the product are
To synthesise 1.0 mole of 2-methylpropan-2-ol from Ethylethanoate ______ equivalents of CH3MgBr reagent will be required. (Integer value)
For distinction between \[\ce{CH3CHO}\] and \[\ce{C6H5CHO}\] the reagent used is ______.
\[\ce{? ->[\Delta, CN-][EtOH, H2O]}\] Benzoin.
The reactant is obtained by dry distillation of the calcium salts of the following pairs:
The major product of the following reaction is:
\[\begin{array}{cc}
\ce{Cl}\phantom{.........................}\\
|\phantom{..........................}\\
\ce{CH3 - CH - CH3 ->[(i) Alc. KOH][(ii) HBr/peroxide (iii) aq. KOH]}
\end{array}\]
Ceric aminonium nitrate and CHCl3/alc. KOH are used for the identification of functional groups present in ______ and ______ respectively.
