Advertisements
Advertisements
Question
Why is the reactivity of all the three classes of alcohols with conc. \[\ce{HCl}\] and \[\ce{ZnCl2}\] (Lucas reagent) different?
Advertisements
Solution
The alcohol combine with \[\ce{HCl}\] to form protonated alcohol. The positive charge on oxygen weakens the C – O bond leading to its cleavage.
(i)
\[\begin{array}{cc}
\phantom{...............}\ce{H}\\
\phantom{...............}|\\
\ce{R - \underset{\bullet\bullet}{\overset{\bullet\bullet}{O}} - H + HCl ⇌[fast] R - \underset{\bullet\bullet}{O}^+ - H + Cl-}
\end{array}\]
(ii)

(iii)
\[\ce{R+ + Cl- ->[fast] RCl}\]
The rate-determining step in the above mechanism is (ii), which is a slow step reaction. The stability of carbocation will determine the reactivity of the reaction. Since the order of stability of carbocation is: Pri. < sec. < tert. Hence the order of formation of alkyl halide in the above reaction is pri. < sec. < tert.
APPEARS IN
RELATED QUESTIONS
How will you convert: Propene to Propan-2-ol?
Name the reagents used in the following reactions:

Write the structures of the main products when acetone (CH3 − CO − CH3) reacts with the following reagents :
CH3MgBr and then H3O+
Show how is the following alcohol prepared by the reaction of a suitable Grignard reagent on methanal?

How is the following conversion carried out?
\[\ce{Benzyl chloride -> Benzyl alcohol}\]

Show how you would synthesise the following alcohol from an appropriate alkene?
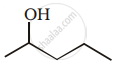
Show how you would synthesise the following alcohol from an appropriate alkene?
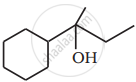
Write the structure of main compounds A and B in the following reaction:
\[\ce{CH3CH2CN->[CH3MgBRH/3O+]A->[LiAIH4]B}\]
Primary alcohols are prepared by the reduction of carboxylic acids. Though lithium aluminium hydride is a strong reducing agent, it is not used in the reaction. This is so because:
Aldehydes are reduced to the corresponding alcohols by the addition of hydrogen in the presence of catalysts to form ____________.
Monochlorination of toluene in sunlight followed by hydrolysis by aq. \[\ce{NaOH}\] yields.
The Wittig reaction is a reaction between a carbonyl compound (aldehyde or ketone only) and a species known as a phosphoniumylide. What is the expected final product in the Wittig reaction?
Carboxylic acids are more acidic than phenol and alcohol because of
A compound does not react with 2, 4 dinitrophenylhydrazine, the compound is:
The reagent used for dehydration of an alcohol is
The best reagent to convert pent - 3 - en - 2 - ol into pent - 3 - en - 2 one is ______.
To synthesise 1.0 mole of 2-methylpropan-2-ol from Ethylethanoate ______ equivalents of CH3MgBr reagent will be required. (Integer value)
Write the mechanism of acid dehydration of ethanol to yield ethene.
