Advertisements
Advertisements
Question
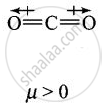
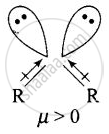
Explain why is \[\ce{O = C =O}\] nonpolar while \[\ce{R - O - R}\] is polar.
Advertisements
Solution
\[\ce{O = C =O}\] molecule is linear so that the polarities of two C – O bonds get cancelled and the molecule is linear.
Ethers have structures similar to water and have angular or bent structure. Therefore, the polarity of two R – O groups does not get cancelled and these have net dipole moment. Thus, \[\ce{R - O - R}\] is polar.
APPEARS IN
RELATED QUESTIONS
Write the structure of the product of the following reaction:

Write the structure of the product of the following reaction:
\[\begin{array}{cc}
\ce{CH3 - CH2 - CH - CHO ->[NaBH4]}\\
|\phantom{....}\\
\ce{CH3}\\
\end{array}\]
Write the structure of the compound whose IUPAC name is as follows:
2-Methylbutan-2-ol
Write the structure of the compound whose IUPAC name is as follows:
3,5-Dimethylhexane-1, 3, 5-triol
Write the structure of the compound whose IUPAC name is as follows:
2,3-Diethylphenol
Write the structure of the compound whose IUPAC name is as follows:
Cyclohexylmethanol
Write the structure of the compound whose IUPAC name is as follows:
3-Cyclohexylpentan-3-ol
Write the structure of the compound whose IUPAC name is as follows:
Cyclopent-3-en-1-ol
Write the structures of the isomers of alcohols with molecular formula \[\ce{C4H10O}\]. Which of these exhibits optical activity?
Cresol is:-
4 – chloro – 3, 5 – dimethyl phenol is called
Glycerol is used
\[\begin{array}{cc}\phantom{......}\ce{OH}\\
\phantom{....}|\\\ce{CH3CH2 - C - CH3}\\
\phantom{....}|\\\phantom{.....}\ce{Ph}\end{array}\] cannot be prepared by ______.
Give IUPAC name of the compound given below.
\[\begin{array}{cc}
\phantom{.}\ce{CH3 - CH - CH2-CH2-CH-CH3}\\
\phantom{.}|\phantom{............................}|\\
\phantom{....}\ce{Cl}\phantom{.........................}\ce{OH}\\
\end{array}\]
Total number of possible isomers (both structural as well as stereoisomers) of cyclic ethers of molecular formula C4H8O is ______.
