Advertisements
Advertisements
प्रश्न
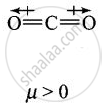
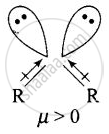
Explain why is \[\ce{O = C =O}\] nonpolar while \[\ce{R - O - R}\] is polar.
Advertisements
उत्तर
\[\ce{O = C =O}\] molecule is linear so that the polarities of two C – O bonds get cancelled and the molecule is linear.
Ethers have structures similar to water and have angular or bent structure. Therefore, the polarity of two R – O groups does not get cancelled and these have net dipole moment. Thus, \[\ce{R - O - R}\] is polar.
APPEARS IN
संबंधित प्रश्न
\[\ce{CH3 - CH = CH2 ->[H2O/H+]}\]
Write the structure of the compound whose IUPAC name is as follows:
2-Methylbutan-2-ol
Write the structure of the compound whose IUPAC name is as follows:
1-Phenylpropan-2-ol
Write the structure of the compound whose IUPAC name is as follows:
1-Ethoxypropane
Write the structure of the compound whose IUPAC name is as follows:
Cyclohexylmethanol
Write the structure of the compound whose IUPAC name is as follows:
3-Cyclohexylpentan-3-ol
What is the structure and IUPAC name of glycerol?
When phenol is treated with bromine water, white precipitate is obtained. Give the structure and the name of the compound formed.
Write the structures of the isomers of alcohols with molecular formula \[\ce{C4H10O}\]. Which of these exhibits optical activity?
Which of the following does not form phenol or peroxide?
Epoxides are
Cresol is:-
Glycerol is used
Diethyl ether finds are in medicine as
Ether can be used
\[\begin{array}{cc}\phantom{......}\ce{OH}\\
\phantom{....}|\\\ce{CH3CH2 - C - CH3}\\
\phantom{....}|\\\phantom{.....}\ce{Ph}\end{array}\] cannot be prepared by ______.
Why is the C-O bond length in phenols less than that in methanol?
Write the structure of the compound whose IUPAC name is as follows:
4-Chloro-3-ethylbutan-1-ol
Total number of possible isomers (both structural as well as stereoisomers) of cyclic ethers of molecular formula C4H8O is ______.
