Advertisements
Advertisements
Question
How is the following conversion carried out?
\[\ce{Methyl magnesium bromide -> 2-Methylpropan-2-ol}\]
Advertisements
Solution 1
When methyl magnesium bromide is treated with propane, an adduct forms that gives 2-methylpropane-2-ol on hydrolysis.
\[\begin{array}{cc}
\ce{H3C}\phantom{..................................}\\
\backslash\phantom{.............................}\\
\ce{C = O + CH3 - MgBr ->}\phantom{.}\\
/\phantom{............................}\\
\ce{H3C}\phantom{..................................}
\end{array}\]\[\begin{bmatrix}
\ce{CH3}\phantom{.}\\
|\phantom{.....}\\
\ce{CH3 - C - \overset{-}{O}\overset{+}{Mg}Br}\\
|\phantom{.....}\\
\ce{\underset{Adduct}{CH3}}\phantom{.}\\
\end{bmatrix}\]\[\begin{array}{cc}
\ce{OH}\phantom{.........}\\
|\phantom{............}\\
\ce{->[H2O]CH3 - C - CH3 + Mg(OH)Br}\\
|\phantom{.............}\\
\ce{\underset{2-Methylpropan-2-ol}{CH3}}\phantom{..........}\\
\end{array}\]
Solution 2
\[\begin{array}{cc}
\phantom{....................}\ce{O}\phantom{............................}\ce{CH3}\\
\phantom{................}||\phantom{..............................}|\\
\ce{\underset{bromide}{\underset{Methyl magnesium}{H3C - MgBr}} + C ->[(i) Dry ether][(ii) H3O+] CH3 - C - OH}\\
\phantom{..............}/\phantom{.}\backslash\phantom{.............................}|\\
\phantom{....................}\ce{\ce{H3C}\phantom{....}\ce{CH3}\phantom{..............}\underset{2-Methylpropan-2-ol}{CH3}}
\end{array}\]
APPEARS IN
RELATED QUESTIONS
How will you convert: Propene to Propan-2-ol?
Name the reagents used in the following reactions:

How do you convert the following : Propan-2-ol to 2-methylpropan-2-ol
How is the following conversion carried out?
\[\ce{Propene -> Propan-2-ol}\]
How is the following conversion carried out?
\[\ce{Benzyl chloride -> Benzyl alcohol}\]
Name the reagent used in the following reaction:
Butan-2-one to butan-2-ol.

Show how you would synthesise the following alcohol from an appropriate alkene?

Show how you would synthesise the following alcohol from an appropriate alkene?
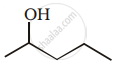
Alkenes convert into alcohols by ____________.
Why is the reactivity of all the three classes of alcohols with conc. \[\ce{HCl}\] and \[\ce{ZnCl2}\] (Lucas reagent) different?
Select the acid(s) which cannot be prepared by Grignard reagent.
Alkaline hydrolysis of an alkyl halide can be preferably carried out using ______.
The best reagent to convert pent - 3 - en - 2 - ol into pent - 3 - en - 2 one is ______.
For distinction between \[\ce{CH3CHO}\] and \[\ce{C6H5CHO}\] the reagent used is ______.
\[\ce{? ->[\Delta, CN-][EtOH, H2O]}\] Benzoin.
The reactant is obtained by dry distillation of the calcium salts of the following pairs:
How are the following conversions carried out?
\[\ce{Methyl magnesium bromide->2-Methylpropan-2-ol}\]
How are the following conversion carried out?
\[\ce{Methyl magnesium bromide -> 2-Methylpropan-2-ol}\]
