Advertisements
Advertisements
Question
How can propan-2-one be converted into tert- butyl alcohol?
Advertisements
Solution
\[\begin{array}{cc}
\phantom{...........}\ce{O}\phantom{................................}\ce{CH3}\phantom{.................}\ce{CH3}\phantom{.....................}\\
\phantom{............}||\phantom{.................................}|\phantom{.....................}|\phantom{.........................}\\
\phantom{}\ce{CH3 - C - CH3 + CH3 MgI ->[Ether] CH3 - C - CH3 ->[H+][H2O] CH3 - C - CH3 + Mg(I)OH}\phantom{.}\\
\phantom{......................}|\phantom{.....................}|\phantom{.}\\
\phantom{.........................}\ce{\underset{Addition product}{CH3}}\phantom{.........}\ce{\underset{\underset{(2-methylpropan-2-ol)}{tert-Butyl alcohol}}{OH}}\phantom{.}
\end{array}\]
APPEARS IN
RELATED QUESTIONS
How will you convert: Propene to Propan-2-ol?
Name the reagents used in the following reactions:

Name the reagents used in the following reactions:

Show how is the following alcohol prepared by the reaction of a suitable Grignard reagent on methanal?
\[\begin{array}{cc}
\ce{CH3 - CH - CH2OH}\\
|\phantom{......}\\
\ce{CH3}\phantom{...}
\end{array}\]
Predict the major product of acid catalysed dehydration of 1-methylcyclohexanol.
Predict the major product of acid catalysed dehydration of butan-1-ol.
How is the following conversion carried out?
\[\ce{Ethyl magnesium chloride -> Propan-1-ol}\]
Show how you would synthesise the following alcohol from an appropriate alkene?

Show how you would synthesise the following alcohol from an appropriate alkene?
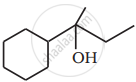
Write the structures of A, B and C in the following reactions :
Ketones react with Grignard reagent to produce ____________.
Primary alcohols are prepared by the reduction of carboxylic acids. Though lithium aluminium hydride is a strong reducing agent, it is not used in the reaction. This is so because:
Benzaldehyde differs from acetaldehyde in that:
Explain a process in which a biocatalyst is used in industrial preparation of a compound known to you.
Carboxylic acids are more acidic than phenol and alcohol because of
Glycerol as a trimester present in
The best reagent to convert pent - 3 - en - 2 - ol into pent - 3 - en - 2 one is ______.
To synthesise 1.0 mole of 2-methylpropan-2-ol from Ethylethanoate ______ equivalents of CH3MgBr reagent will be required. (Integer value)
How are the following conversions carried out?
\[\ce{Methyl magnesium bromide ->2-Methylpropan-2-ol}\]
