Advertisements
Advertisements
प्रश्न

Advertisements
उत्तर
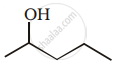
The given alcohols can be synthesized by applying Markovnikov’s rule of acid-catalyzed hydration of appropriate alkenes.
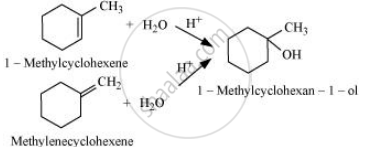
APPEARS IN
संबंधित प्रश्न
Write the main product(s) in each of the following reactions:

What is meant by hydroboration-oxidation reaction? Illustrate it with an example.
How is the following conversion carried out?
\[\ce{Benzyl chloride -> Benzyl alcohol}\]
How is the following conversion carried out?
\[\ce{Methyl magnesium bromide → 2-Methylpropan-2-ol}\]
Name the reagent used in the following reaction:
Butan-2-one to butan-2-ol.
Show how you would synthesise the following alcohol from an appropriate alkene?
Monochlorination of toluene in sunlight followed by hydrolysis by aq. \[\ce{NaOH}\] yields.
Benzaldehyde differs from acetaldehyde in that:
Alkenes convert into alcohols by ____________.
Which of the following reacts with NaOH to give alcohol?
Identify ‘C’ in the following:

Why is the reactivity of all the three classes of alcohols with conc. \[\ce{HCl}\] and \[\ce{ZnCl2}\] (Lucas reagent) different?
Magnesium wire continues to burn in the atmosphere of CO2 because
Carboxylic acids are more acidic than phenol and alcohol because of
The reagent used for dehydration of an alcohol is
When alcohol react with concentrated H2SO4 intermediate compound formed is
Alkaline hydrolysis of an alkyl halide can be preferably carried out using ______.
The major product of acid catalysed dehydration of 1-methylcyclohexanol is ______.
\[\ce{C3H8O ->[{[O]}][K2Cr2O7/H2SO4] C3H6O ->[I2 + NaOH(aq.)] CHI3}\]
In this reaction the first compound is:
For distinction between \[\ce{CH3CHO}\] and \[\ce{C6H5CHO}\] the reagent used is ______.
