Advertisements
Advertisements
प्रश्न
When 3-methylbutan-2-ol is treated with HBr, the following reaction takes place:
\[\begin{array}{cc}
\phantom{.......................}\ce{Br}\\
\phantom{......................}|\\
\ce{CH3 - CH - CH - CH3 ->[HBr] CH3 - C - CH2 - CH3}\\
\phantom{.}|\phantom{......}|\phantom{......................}|\phantom{........}\\
\phantom{}\ce{CH3}\phantom{...}\ce{OH}\phantom{...................}\ce{CH3}\phantom{.....}
\end{array}\]
Give a mechanism for this reaction.
(Hint: The secondary carbocation formed in step II rearranges to a more
stable tertiary carbocation by a hydride ion shift from 3rd carbon atom.)
Advertisements
उत्तर
The described reaction is an example of carbocation rearrangement that occurs via hydride shift. The mechanism for it is –
Step 1: Formation of carbocation: Protonation of alcohol.
\[\ce{HBr -> H+ + B\overset{—}{r}}\]
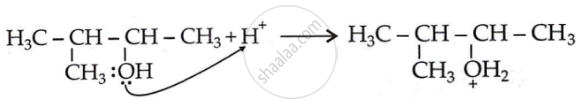
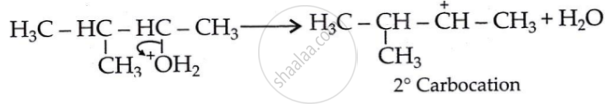
Step 2: 1, 2-hydride shift: Formation of a more stable, 3° carbocation.
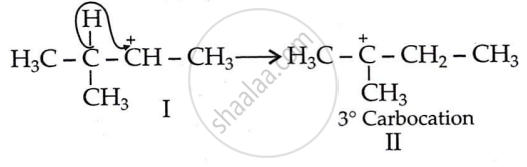
Initially, a 2° carbocation (I) was produced. However, the more stable 3° counterpart causes a hydride shift, forming the more stable carbocation (II).
Step 3: Attack of nucleophile: Generation of product.
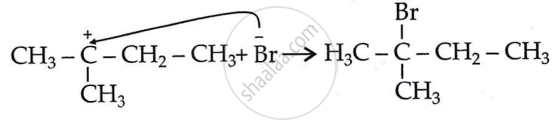
APPEARS IN
संबंधित प्रश्न
Write the final product(s) in each of the following reactions:

Give reasons for the following:
o-nitrophenol is more acidic than o-methoxyphenol.
Write the mechanism of the following reaction :

Give the equation of the following reaction:
Oxidation of propan-1-ol with alkaline KMnO4 solution.
Name the reagent used in the following reaction:
Benzyl alcohol to benzoic acid.
Write the mechanism (using curved arrow notation) of the following reaction :
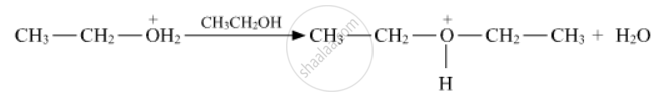
The compound which reacts fastest with Lucas reagent at room temperature is:
In the reduction \[\ce{R - CHO + H2 -> RCH2OH}\] the catalyst used is:
Which of the following are used to convert RCHO into RCH2OH?
(i) H2/Pd
(ii) LiAlH4
(iii) NaBH4
(iv) Reaction with RMgX followed by hydrolysis
Which of the following is not true in case of reaction with heated copper at 300°C?
Explain why is OH group in phenols more strongly held as compared to OH group in alcohols.
What is Lucas reagent?
What happens when (CH3)3 C – OH is heated with Cu/573 K?
Write the chemical equation in support of your answer.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
