Advertisements
Advertisements
Question
How will you convert: Phenol to 2, 4, 6 − trinitrophenol?
Advertisements
Solution
When concentrated nitric acid is added to phenol in the presence of sulphuric acid it gives 2, 4, 6-trinitrophenol.

APPEARS IN
RELATED QUESTIONS
How will you convert: Propene to Propan-2-ol?
Write the structures of the main products when acetone (CH3 − CO − CH3) reacts with the following reagents :
CH3MgBr and then H3O+
What is meant by hydroboration-oxidation reaction? Illustrate it with an example.
Write the mechanism of hydration of ethene to yield ethanol.
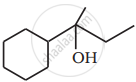
Show how you would synthesise the following alcohol from an appropriate alkene?
Aldehydes are reduced to the corresponding alcohols by the addition of hydrogen in the presence of catalysts to form ____________.
When alcohol react with concentrated H2SO4 intermediate compound formed is
Glycerol as a trimester present in
\[\ce{C3H8O ->[{[O]}][K2Cr2O7/H2SO4] C3H6O ->[I2 + NaOH(aq.)] CHI3}\]
In this reaction the first compound is:
How are the following conversions carried out?
\[\ce{Methyl magnesium bromide ->2-Methylpropan-2-ol}\]
