Advertisements
Advertisements
Question
Write the mechanism of hydration of ethene to yield ethanol.
Advertisements
Solution 1
Step 1: Protonation of the alkene to form a carbocation by the electrophilic attack of H3O+.
\[\ce{H2O + H+ -> H3O+}\]
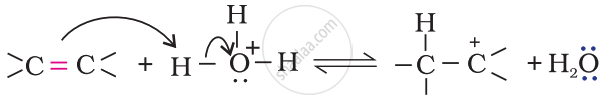
Step 2: Nucleophilic attack of water on carbocation.
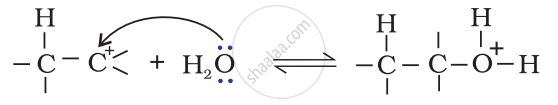
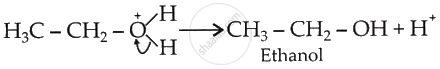
Step 3: Deprotonation to form an alcohol.

Solution 2
The acid-catalysed hydration of ethene may be represented as:
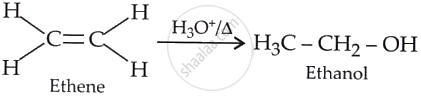
The mechanism followed by the above reaction may be depicted as:
Generation of carbocation (Step 1): Protonation of ethene yields a carbocation as,
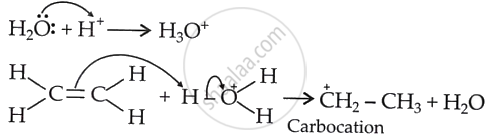
Nucleophilic attack of water on carbocation (Step 2): The carbocation polarises the lone pair electrons of O of water, and hydration takes place.
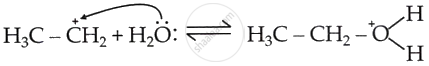
Formation of product, regeneration of the proton (Step 3):
APPEARS IN
RELATED QUESTIONS
Write the main product(s) in each of the following reactions:

Show how is the following alcohol prepared by the reaction of a suitable Grignard reagent on methanal?

How is the following conversion carried out?
\[\ce{Propene -> Propan-2-ol}\]
How is the following conversion carried out?
\[\ce{Ethyl magnesium chloride -> Propan-1-ol}\]
Show how you would synthesise the following alcohol from an appropriate alkene?

How will you convert: Phenol to 2, 4, 6 − trinitrophenol?
Name the reagents used in the following reactions:

Ketones react with Grignard reagent to produce ____________.
Monochlorination of toluene in sunlight followed by hydrolysis by aq. \[\ce{NaOH}\] yields.
Benzaldehyde differs from acetaldehyde in that:
Alkenes convert into alcohols by ____________.
The Wittig reaction is a reaction between a carbonyl compound (aldehyde or ketone only) and a species known as a phosphoniumylide. What is the expected final product in the Wittig reaction?
Select the acid(s) which cannot be prepared by Grignard reagent.
The reagent used for dehydration of an alcohol is
Alkaline hydrolysis of an alkyl halide can be preferably carried out using ______.
For distinction between \[\ce{CH3CHO}\] and \[\ce{C6H5CHO}\] the reagent used is ______.
Given below are two statements:
Statement I: On heating with KHSO4, glycerol is dehydrated and acrolein is formed.
Statement II: Acrolein has a fruity odour and can be used to test glycerol's presence.
Choose the correct option.
Ceric aminonium nitrate and CHCl3/alc. KOH are used for the identification of functional groups present in ______ and ______ respectively.
