Advertisements
Advertisements
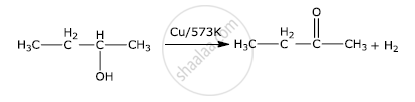
प्रश्न
Write the final product(s) in each of the following reactions:

Advertisements
उत्तर
APPEARS IN
संबंधित प्रश्न
Write the mechanism of the following reaction :

Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene.
Name the reagent used in the following reaction:
Benzyl alcohol to benzoic acid.
Identify the secondary alcohols from the following set:
- \[\ce{CH3CH2CH(OH)CH3}\]
- \[\ce{(C2H5)3COH}\]
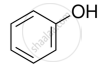
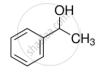
Name the factors responsible for the solubility of alcohols in water.
Why is the C – O – H bond angle in alcohols slightly less than the tetrahedral angle whereas the C – O – C bond angle in ether is slightly greater?
What is Lucas reagent?
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene.
