Advertisements
Advertisements
Question
For a chemical reaction R → P, the variation in the concentration (R) vs. time (t) plot is given as:

(i) Predict the order of the reaction.
(ii) What is the slope of the curve?
(iii) Write the unit of rate constant for this reaction.
Advertisements
Solution 1
For a chemical reaction R→P, the variation in the concentration (R) vs. time (t) plot is given as follows:

(i) The order of the reaction zero.
(ii) Slope = -k
Solution 2
(i) Predict the order of the reaction
The graph shows a straight line when concentration of reactant R is plotted against time t.
[R] = [R]0 − kt
Since the plot is a straight line with a negative slope, the reaction is zero order.
(ii) Slope of the curve
From the zero-order equation:
[R] = [R]0 − kt
The slope of the [R] vs t graph is −k (negative of the rate constant).
(iii) Unit of rate constant
For a zero-order reaction:
Rate = k
Rate has units of concentration/time.
If concentration is in mol L−1 and time in seconds:
Unit of k = mol L−1s−1
APPEARS IN
RELATED QUESTIONS
A → B is a first order reaction with rate 6.6 × 10-5m-s-1. When [A] is 0.6m, rate constant of the reaction is
- 1.1 × 10-5s-1
- 1.1 × 10-4s-1
- 9 × 10-5s-1
- 9 × 10-4s-1
In a reaction between A and B, the initial rate of reaction (r0) was measured for different initial concentrations of A and B as given below:
| A/mol L−1 | 0.20 | 0.20 | 0.40 |
| B/mol L−1 | 0.30 | 0.10 | 0.05 |
| r0/mol L−1 s−1 | 5.07 × 10−5 | 5.07 × 10−5 | 1.43 × 10−4 |
What is the order of the reaction with respect to A and B?
How does calcination differ from roasting?
Define the following term:
Pseudo first-order reaction
Which of the following statements is not correct about order of a reaction.
Compounds ‘A’ and ‘B’ react according to the following chemical equation.
\[\ce{A(g) + 2B(g) -> 2C(g)}\]
Concentration of either ‘A’ or ‘B’ were changed keeping the concentrations of one of the reactants constant and rates were measured as a function of initial concentration. Following results were obtained. Choose the correct option for the rate equations for this reaction.
| Experiment | Initial concentration of [A]/mol L–¹ |
Initial concentration of [B]/mol L–¹ |
Initial rate of formation of [C]/mol L–¹ s–¹ |
| 1. | 0.30 | 0.30 | 0.10 |
| 2. | 0.30 | 0.60 | 0.40 |
| 3. | 0.60 | 0.30 | 0.20 |
For a general reaction A → B, plot of concentration of A vs time is given in figure. Answer the following question on the basis of this graph.
(i) What is the order of the reaction?
(ii) What is the slope of the curve?
(iii) What are the units of rate constant?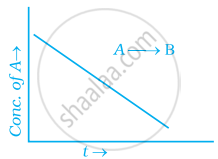
Match the graph given in Column I with the order of reaction given in Column II. More than one item in Column I may link to the same item of Column II.
| Column I | Column II | |
| (i) |  |
|
| (ii) |  |
(a) 1st order |
| (iii) |  |
(b) Zero-order |
| (iv) |  |
Identify the order of reaction from the following unit for its rate constant:
L mol–1 s–1
Higher yield of NO in \[\ce{N2(g) + O2 <=> 2NO(g)}\] can be obtained at:
[ΔH of the reaction = +180.7 kJ mol−1]
- higher temperature
- lower temperature
- higher concentration of N2
- higher concentration of O2
Choose the correct answer from the options given below:
