Advertisements
Advertisements
Question
In a reaction between A and B, the initial rate of reaction (r0) was measured for different initial concentrations of A and B as given below:
| A/mol L−1 | 0.20 | 0.20 | 0.40 |
| B/mol L−1 | 0.30 | 0.10 | 0.05 |
| r0/mol L−1 s−1 | 5.07 × 10−5 | 5.07 × 10−5 | 1.43 × 10−4 |
What is the order of the reaction with respect to A and B?
Advertisements
Solution
Suppose the order of the reaction with respect to A is m and with respect to B is n.
∴ Rate (r0) = k[A]m [B]n
For the given data, we have,
Rate (r1) = 5.07 × 10−5
= k (0.20)m × (0.30)n ...(i)
Rate (r2) = 5.07 × 10−5
= k (0.20)m × (0.10)n ...(ii)
Rate (r3) = 1.43 × 10−4
= k (0.40)m × (0.05)n ...(iii)
Dividing eq. (i) by eq. (ii), we have,
`(5.07 xx 10^-5)/(5.07 xx 10^-5) = (k (0.20)^m xx (0.30)^n)/(k (0.20)^m xx (0.10)^n)`
⇒ 1 = (3)n
⇒ n = 0
Dividing eq. (iii) by eq. (ii) and putting the value of n, we get,
`(1.43 xx 10^-4)/(5.07 xx 10^-5) = (k (0.40)^m xx (0.05)^0)/(k (0.20)^m xx (0.10)^0)`
⇒ `(1.43 xx 10^-4)/(5.07 xx 10^-5)` = 2m
⇒ (2)m = 2.82
⇒ m log10 2 = log10 2.82 = 0.45
⇒ m = `0.45/(log_10 2)`
= `0.45/0.3010`
= 1.49
= 1.50
Hence, the order with respect to A is 1.5, and the order with respect to B is 0.
APPEARS IN
RELATED QUESTIONS
Define “zero order reaction”.
Write molecularity of the following reaction:
2NO(g)+O2(g)→2NO2(g)
For a reaction A + B ⟶ P, the rate is given by
Rate = k [A] [B]2
How is the rate of reaction affected if the concentration of B is doubled?
A reaction is second order with respect to a reactant. How is the rate of reaction affected if the concentration of the reactant is doubled?
A reaction is first order in A and second order in B. Write the differential rate equation.
For a reaction R ---> P, half-life (t1/2) is observed to be independent of the initial concentration of reactants. What is the order of reaction?
Write resonating structures of ozone.
Rate of reaction for the combustion of propane is equal to:
\[\ce{C3H8_{(g)} + 5O2_{(g)} -> 3CO2_{(g)} + 4H2O_{(g)}}\]
For a general reaction A → B, plot of concentration of A vs time is given in figure. Answer the following question on the basis of this graph.
(i) What is the order of the reaction?
(ii) What is the slope of the curve?
(iii) What are the units of rate constant?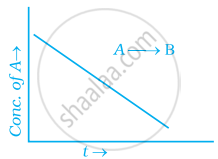
Why does the rate of any reaction generally decreases during the course of the reaction?
Why can’t molecularity of any reaction be equal to zero?
Why can we not determine the order of a reaction by taking into consideration the balanced chemical equation?
Assertion: The enthalpy of reaction remains constant in the presence of a catalyst.
Reason: A catalyst participating in the reaction, forms different activated complex and lowers down the activation energy but the difference in energy of reactant and product remains the same.
The rate constant for the reaction \[\ce{2H2O5 -> 4NO2 + O2}\] is 30 × 10–5 sec–1. if the rate is 204 × 10–5 mol L–1 S–1, then the concentration of N2O5 (in mol–1) is-
For reaction 2A + B → BC + D which of the following does not Express the reaction rates
At concentration of 0.1 and 0.2 mol L–1 the rates of deem position of a compound were found to be 0.18 and 0.72 mol L–1 m–1. What is the order of the reaction?
For a chemical reaction starting with some initial concentration of reactant At as a function of time (t) is given by the equation,
`1/("A"_"t"^4) = 2 + 1.5 xx 10^-3` t
The rate of disappearance of [A] is ____ × 10-2 M/sec when [A] = 2 M.
[Given: [At] in M and t in sec.]
[Express your answer in terms of 10-2 M /s]
[Round off your answer if required]
A flask contains a mixture of compounds A and B. Both compounds decompose by first-order kinetics. The half-lives for A and B are 300 s and 180 s, respectively. If the concentrations of A and B are equal initially, the time required for the concentration of A to be four times that of B (in s) is ______. (Use ln 2 = 0.693)
Which of the following statement is true?
Higher yield of NO in \[\ce{N2(g) + O2 <=> 2NO(g)}\] can be obtained at:
[ΔH of the reaction = +180.7 kJ mol−1]
- higher temperature
- lower temperature
- higher concentration of N2
- higher concentration of O2
Choose the correct answer from the options given below:
