Advertisements
Advertisements
Questions
Identify the order of reaction from the following unit for its rate constant:
L mol–1 s–1
State the order of reaction whose rate constant possess the following unit:
mol–1 L s–1
Identify the order of the reaction if the unit of its rate constant is mol–1 L s–1.
Advertisements
Solution
The unit L mol–1sec–1 for the rate constant is the unit of a second-order reaction.
APPEARS IN
RELATED QUESTIONS
Write molecularity of the following reaction:
2NO(g)+O2(g)→2NO2(g)
For a reaction R ---> P, half-life (t1/2) is observed to be independent of the initial concentration of reactants. What is the order of reaction?
Write the principle behind the following methods of refining:
Hydraulic washing
Consider a first order gas phase decomposition reaction given below :
\[\ce{A(g) -> B(g) + C(g)}\]
The initial pressure of the system before decomposition of A was pi. After lapse of time ‘t’, total pressure of the system increased by x units and became ‘pt’ The rate constant k for the reaction is given as ______.
For a general reaction A → B, plot of concentration of A vs time is given in figure. Answer the following question on the basis of this graph.
(i) What is the order of the reaction?
(ii) What is the slope of the curve?
(iii) What are the units of rate constant?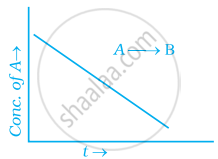
Assertion: Order and molecularity are same.
Reason: Order is determined experimentally and molecularity is the sum of the stoichiometric coefficient of rate determining elementary step.
Assertion: Rate constants determined from Arrhenius equation are fairly accurate for simple as well as complex molecules.
Reason: Reactant molecules undergo chemical change irrespective of their orientation during collision.
For reaction 2A + B → BC + D which of the following does not Express the reaction rates
For the reaction, \[\ce{A +2B → AB2}\], the order w.r.t. reactant A is 2 and w.r.t. reactant B. What will be change in rate of reaction if the concentration of A is doubled and B is halved?
The conversion of molecules A to B follow second order kinetics. If concentration of A is increased to three times, how will it affect the rate of formation of B?
