Advertisements
Advertisements
Question
Consider a first order gas phase decomposition reaction given below :
\[\ce{A(g) -> B(g) + C(g)}\]
The initial pressure of the system before decomposition of A was pi. After lapse of time ‘t’, total pressure of the system increased by x units and became ‘pt’ The rate constant k for the reaction is given as ______.
Options
`k = 2.303/t log p_i/(p_i - x)`
`k = 2.303/t log p_i/(2p_i - p_t)`
`k = 2.303/t log p_i/(2p_i + p_t)`
`k = 2.303/t log p_i/(p_i + x)`
Advertisements
Solution
The initial pressure of the system before decomposition of A was pi. After lapse of time ‘t’, total pressure of the system increased by x units and became ‘pt’ The rate constant k for the reaction is given as `k = 2.303/t log p_i/(2p_i - p_t)`.
Explanation:
Consider a first order gas phase decomposition reaction:
\[\ce{A_{(g)} -> B_{(g)} + C_{(g)}}\]
The initial pressure of the system before decomposition of A was Pi.
After lapse of time (t), total pressure of the system increased by x units and became 'Pt'.
In other words, the pressure of A decreased by x atom.
\[\ce{A_{(g)} -> B_{(g)} + C_{(g)}}\]
Initial pressure: Pi atm
Pressure after time t: (Pi – x) atm x atm x atm
Pt = (Pi – x) + x + x = Pi + x atm
x = Pt – Pi
PA = Pressure of A after time t = Pi - x = Pi - (Pi - Pi) = 2Pi - Pt
`k = 2.303/t log (["A"]_0)/(["A"])`
= `k = 2.303/t log p_i/(2p_i - p_t)`
APPEARS IN
RELATED QUESTIONS
In a first order reaction x → y, 40% of the given sample of compound remains unreacted in 45 minutes. Calculate rate constant of the reaction.
Write molecularity of the following reaction:
2NO(g)+O2(g)→2NO2(g)
The conversion of molecules X to Y follows second order kinetics. If concentration of X is increased to three times how will it affect the rate of formation of Y?
From the rate expression for the following reaction, determine the order of reaction and the dimension of the rate constant.
\[\ce{H2O2_{( aq)} + 3I^-_{( aq)} + 2H^+ -> 2H2O_{(l)} + I^-_3}\] Rate = k[H2O2][I−]
From the rate expression for the following reaction, determine the order of reaction and the dimension of the rate constant.
\[\ce{C2H5Cl_{(g)} -> C2H4_{(g)} + HCl_{(g)}}\] Rate = k [C2H5Cl]
For the reaction:
\[\ce{2A + B -> A2B}\]
the rate = k[A][B]2 with k = 2.0 × 10−6 mol−2 L2 s−1. Calculate the initial rate of the reaction when [A] = 0.1 mol L−1, [B] = 0.2 mol L−1. Calculate the rate of reaction after [A] is reduced to 0.06 mol L−1.
Mention the factors that affect the rate of a chemical reaction.
A reaction is first order in A and second order in B. How is the rate affected on increasing the concentration of B three times?
How does calcination differ from roasting?
Write the principle behind the following methods of refining:
Hydraulic washing
What is the order of a reaction which has a rate expression; Rate = `"k"["A"]^(3/2)["B"]^1`?
Consider the reaction A ⇌ B. The concentration of both the reactants and the products varies exponentially with time. Which of the following figures correctly describes the change in concentration of reactants and products with time?
In a reaction if the concentration of reactant A is tripled, the rate of reaction becomes twenty seven times. What is the order of the reaction?
For a general reaction A → B, plot of concentration of A vs time is given in figure. Answer the following question on the basis of this graph.
(i) What is the order of the reaction?
(ii) What is the slope of the curve?
(iii) What are the units of rate constant?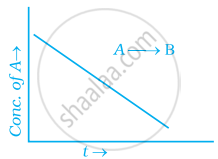
The rate constant for the reaction \[\ce{2H2O5 -> 4NO2 + O2}\] is 30 × 10–5 sec–1. if the rate is 204 × 10–5 mol L–1 S–1, then the concentration of N2O5 (in mol–1) is-
For reaction 2A + B → BC + D which of the following does not Express the reaction rates
At concentration of 0.1 and 0.2 mol L–1 the rates of deem position of a compound were found to be 0.18 and 0.72 mol L–1 m–1. What is the order of the reaction?
The number of molecules of the reactants taking part in a single step of the reaction is indicative of ______.
Read the following passage and answer the questions that follow:
|
The rate of reaction is concerned with decrease in the concentration of reactants or increase in the concentration of products per unit of time. It can be expressed as instantaneous rate at a particular instant of time and average rate over a large interval of time. A number of factors such as temperature, concentration of reactants, catalyst affect the rate of reaction. Mathematical representation of rate of a reaction is given by rate law: Rate = k[A]x [B]y x and y indicate how sensitive the rate is to change in concentration of A and B. Sum of x + y gives the overall order of a reaction. |
- What is the effect of temperature on the rate constant of a reason? [1]
- For a reaction \[\ce{A + B → Product}\], the rate law is given by, Rate = k[A]2 [B]1/2. What is the order of the reaction? [1]
- How order and molecularity are different for complex reactions? [1]
- A first-order reaction has a rate constant 2 × 10–3 s–1. How long will 6 g of this reactant take to reduce to 2 g? [2]
OR
The half-life for radioactive decay of 14C is 6930 years. An archaeological artifact containing wood had only 75% of the 14C found in a living tree. Find the age of the sample.
[log 4 = 0.6021, log 3 = 0.4771, log 2 = 0.3010, log 10 = 1] [2]
