Advertisements
Advertisements
Question
Consider figure and mark the correct option.
Options
Activation energy of forward reaction is E1 + E2 and product is less stable than reactant.
Activation energy of forward reaction is E1 + E2 and product is more stable than reactant.
Activation energy of both forward and backward reaction is E1 + E2 and reactant is more stable than product.
Activation energy of backward reaction is E1 and product is more stable than reactant.
Advertisements
Solution
Activation energy of forward reaction is E1 + E2 and product is less stable than reactant.
Explanation:
Ea (forward) = E1 + E2
Since energy of reactants is less than products and the product is less stable than the reactant.
APPEARS IN
RELATED QUESTIONS
(b) Rate constant ‘k’ of a reaction varies with temperature ‘T’ according to the equation:
`logk=logA-E_a/2.303R(1/T)`
Where Ea is the activation energy. When a graph is plotted for `logk Vs. 1/T` a straight line with a slope of −4250 K is obtained. Calculate ‘Ea’ for the reaction.(R = 8.314 JK−1 mol−1)
The rate of the chemical reaction doubles for an increase of 10 K in absolute temperature from 298 K. Calculate Ea.
The rate constant for the decomposition of N2O5 at various temperatures is given below:
| T/°C | 0 | 20 | 40 | 60 | 80 |
| 105 × k/s−1 | 0.0787 | 1.70 | 25.7 | 178 | 2140 |
Draw a graph between ln k and `1/T` and calculate the values of A and Ea. Predict the rate constant at 30º and 50ºC.
In the Arrhenius equation for a first order reaction, the values of ‘A’ of ‘Ea’ are 4 × 1013 sec−1 and 98.6 kJ mol−1 respectively. At what temperature will its half life period be 10 minutes?
[R = 8.314 J K−1 mol−1]
Explain the following terms :
Half life period of a reaction (t1/2)
Predict the main product of the following reactions: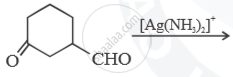
The chemical reaction in which reactants require high amount of activation energy are generally ____________.
Mark the incorrect statements:
(i) Catalyst provides an alternative pathway to reaction mechanism.
(ii) Catalyst raises the activation energy.
(iii) Catalyst lowers the activation energy.
(iv) Catalyst alters enthalpy change of the reaction.
Why does the rate of a reaction increase with rise in temperature?
The activation energy in a chemical reaction is defined as ______.
The activation energy in a chemical reaction is defined as ______.
Explain how and why will the rate of reaction for a given reaction be affected when the temperature at which the reaction was taking place is decreased.
The decomposition of N2O into N2 and O2 in the presence of gaseous argon follows second-order kinetics, with k = (5.0 × 1011 L mol−1 s−1) `"e"^(-(29000 "K")/"T")`. Arrhenius parameters are ______ kJ mol−1.
An exothermic reaction X → Y has an activation energy 30 kJ mol-1. If energy change ΔE during the reaction is - 20 kJ, then the activation energy for the reverse reaction in kJ is ______.
What happens to the rate constant k and activation energy Ea as the temperature of a chemical reaction is increased? Justify.
It is generally observed that the rate of a chemical reaction becomes double with every 10°C rise in temperature. If the generalisation holds true for a reaction in the temperature range of 298 K to 308 K, what would be the value of activation energy (Ea) for the reaction?
Assertion (A): A reaction can have zero activation energy.
Reason (R): The minimum extra amount of energy absorbed by reactant molecules so that their energy becomes equal to the threshold value is called activation energy.
In the light of the above statements, choose the correct answer from the options given below:
