Advertisements
Advertisements
Question
(b) Rate constant ‘k’ of a reaction varies with temperature ‘T’ according to the equation:
`logk=logA-E_a/2.303R(1/T)`
Where Ea is the activation energy. When a graph is plotted for `logk Vs. 1/T` a straight line with a slope of −4250 K is obtained. Calculate ‘Ea’ for the reaction.(R = 8.314 JK−1 mol−1)
Advertisements
Solution
`log k=logA-E_a/2.303R(1/T)`
Ea → Activation energy
The above equation is like y = mx + c where if we plot y v/s x we get a straight line with slope ‘m’ and intercept ‘c’.
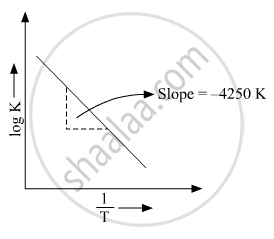
So, slope is equal to`=-E_a/2.303R`
`-E_a/2.303R=-4250k=>E_a=4250 xx 2.303 xx 8.314 =81,375.3535 j mol^(-1)`
`E_a=81.3753 KJ mol^(-1)`
APPEARS IN
RELATED QUESTIONS
The rate constant for the first-order decomposition of H2O2 is given by the following equation:
`logk=14.2-(1.0xx10^4)/TK`
Calculate Ea for this reaction and rate constant k if its half-life period be 200 minutes.
(Given: R = 8.314 JK–1 mol–1)
What will be the effect of temperature on rate constant?
The activation energy for the reaction \[\ce{2 HI_{(g)} -> H2_{(g)} + I2_{(g)}}\] is 209.5 kJ mol−1 at 581K. Calculate the fraction of molecules of reactants having energy equal to or greater than activation energy?
Define activation energy.
During decomposition of an activated complex:
(i) energy is always released
(ii) energy is always absorbed
(iii) energy does not change
(iv) reactants may be formed
Oxygen is available in plenty in air yet fuels do not burn by themselves at room temperature. Explain.
What happens to most probable kinetic energy and the energy of activation with increase in temperature?
Total number of vibrational degrees of freedom present in CO2 molecule is
Arrhenius equation can be represented graphically as follows:

The (i) intercept and (ii) slope of the graph are:
A schematic plot of ln Keq versus inverse of temperature for a reaction is shown below

The reaction must be:
