Advertisements
Advertisements
Question
Predict the main product of the following reactions: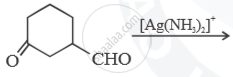
Advertisements
Solution
Predict main product of following reaction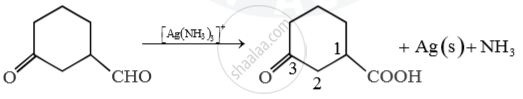
Main product 3-oxocyclohexanecarboxylic acid
APPEARS IN
RELATED QUESTIONS
Explain a graphical method to determine activation energy of a reaction.
The activation energy for the reaction \[\ce{2 HI_{(g)} -> H2_{(g)} + I2_{(g)}}\] is 209.5 kJ mol−1 at 581 K. Calculate the fraction of molecules of reactants having energy equal to or greater than activation energy?
The decomposition of A into product has value of k as 4.5 × 103 s−1 at 10°C and energy of activation 60 kJ mol−1. At what temperature would k be 1.5 × 104 s−1?
A first-order reaction is 50% completed in 40 minutes at 300 K and in 20 minutes at 320 K. Calculate the activation energy of the reaction. (Given : log 2 = 0·3010, log 4 = 0·6021, R = 8·314 JK–1 mol–1)
Explain the following terms :
Half life period of a reaction (t1/2)
The chemical reaction in which reactants require high amount of activation energy are generally ____________.
The rate of chemical reaction becomes double for every 10° rise in temperature because of ____________.
A first-order reaction is 50% complete in 30 minutes at 300 K and in 10 minutes at 320 K. Calculate activation energy (Ea) for the reaction. [R = 8.314 J K−1 mol−1]
[Given: log 2 = 0.3010, log 3 = 0.4771, log 4 = 0.6021]
What happens to the rate constant k and activation energy Ea as the temperature of a chemical reaction is increased? Justify.
Which plot of ln k vs `1/T` is consistent with the Arrhenius equation?
