Advertisements
Advertisements
Question
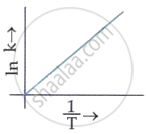
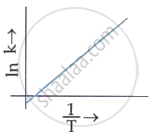
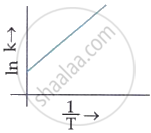
Which plot of ln k vs `1/T` is consistent with the Arrhenius equation?
Options
MCQ
Advertisements
Solution
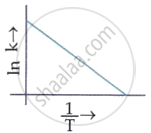
Explanation:
The Arrhenius equation is:
k = `Ae ^(-E_a//RT`
In (k) = `ln (A) + ln(e ^(-E_a//RT))`
In (k) = `ln (A) + (-E_a//RT)`
ln (k) = `ln (A) + (-E_a//R) (1/T)`
Since ln (A) is a constant, the equation corresponds to that of a straight line (y = mx + c) whose slope (m) is `-E_a/R`. When the logarithm of the rate constant (ln K) is plotted on the Y-axis and the inverse of the absolute temperature `(1/T)` is plotted on the X-axis, the resulting graph is called an Arrhenius plot.
shaalaa.com
Is there an error in this question or solution?