Advertisements
Advertisements
Question
A first-order reaction is 50% completed in 40 minutes at 300 K and in 20 minutes at 320 K. Calculate the activation energy of the reaction. (Given : log 2 = 0·3010, log 4 = 0·6021, R = 8·314 JK–1 mol–1)
Advertisements
Solution
Given
t1/2 = 40 min at temperature (T1) = 300 K
t1/2 = 20 min at temperature (T2) = 320 K
t1/2 = 40 min, t1/2 = 20 min
`k_1 = 0.693/40`
`k_2 = 0.693/20`
According to Arrhenius equation
`log (k_2/k_1) = "E"_"a"/(2.303 " R") [1/"T"_1 - 1/"T"_2]`
`= "E"_"a"/(2.303 " R") [("T"_2 - "T"_1)/("T"_1"T"_2)]`
`log ((0.0693/20)/(0.0693/40)) = "E"_"a"/(2.303 xx 8.314) [(320 - 300)/(300 xx 320)]`
`therefore 0.3010 = "E"_"a"/19.147 [0.0002083]`
Ea = 27664 J/mol
Ea = 27.7 kJ/mol
APPEARS IN
RELATED QUESTIONS
Explain a graphical method to determine activation energy of a reaction.
What will be the effect of temperature on rate constant?
The rate constant for the decomposition of N2O5 at various temperatures is given below:
| T/°C | 0 | 20 | 40 | 60 | 80 |
| 105 × k/s−1 | 0.0787 | 1.70 | 25.7 | 178 | 2140 |
Draw a graph between ln k and `1/T` and calculate the values of A and Ea. Predict the rate constant at 30º and 50ºC.
Define activation energy.
Predict the main product of the following reactions: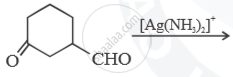
The chemical reaction in which reactants require high amount of activation energy are generally ____________.
The rate of chemical reaction becomes double for every 10° rise in temperature because of ____________.
The activation energy of one of the reactions in a biochemical process is 532611 J mol–1. When the temperature falls from 310 K to 300 K, the change in rate constant observed is k300 = x × 10–3 k310. The value of x is ______.
[Given: ln 10 = 2.3, R = 8.3 J K–1 mol–1]
The rate of a reaction quadruples when temperature changes from 27°C to 57°C calculate the energy of activation.
(Given: R = 8. 314 J K−1 mol−1, log 4 = 0.6021)
Given below are two statements:
Statement I: The nutrient deficient water bodies lead to eutrophication.
Statement II: Eutrophication leads to decrease in the level of oxygen in the water bodies.
In the light of the above statements, choose the correct answer from the options given below:
