Advertisements
Advertisements
प्रश्न
Predict the main product of the following reactions: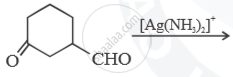
Advertisements
उत्तर
Predict main product of following reaction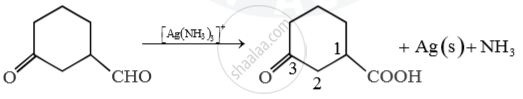
Main product 3-oxocyclohexanecarboxylic acid
APPEARS IN
संबंधित प्रश्न
The rate constant of a first order reaction increases from 2 × 10−2 to 4 × 10−2 when the temperature changes from 300 K to 310 K. Calculate the energy of activation (Ea).
(log 2 = 0.301, log 3 = 0.4771, log 4 = 0.6021)
Explain the following terms :
Half life period of a reaction (t1/2)
The rate of chemical reaction becomes double for every 10° rise in temperature because of ____________.
Oxygen is available in plenty in air yet fuels do not burn by themselves at room temperature. Explain.
What happens to most probable kinetic energy and the energy of activation with increase in temperature?
For an endothermic reaction energy of activation is Ea and enthalpy of reaction ΔH (both of there in KJ moI–1) minimum value of Ea will be ______.
In respect of the eqn k = \[\ce{Ae^{{-E_a}/{RT}}}\] in chemical kinetics, which one of the following statement is correct?
The slope of Arrhenius Plot `("In" "k" "v"//"s" 1/"T")` of first-order reaction is −5 × 103 K. The value of Ea of the reaction is. Choose the correct option for your answer. [Given R = 8.314 JK−1mol−1]
A schematic plot of ln Keq versus inverse of temperature for a reaction is shown below

The reaction must be:
It is generally observed that the rate of a chemical reaction becomes double with every 10°C rise in temperature. If the generalisation holds true for a reaction in the temperature range of 298 K to 308 K, what would be the value of activation energy (Ea) for the reaction?
