Advertisements
Advertisements
Question
Match the graph given in Column I with the order of reaction given in Column II. More than one item in Column I may link to the same item of Column II.
| Column I | Column II | |
| (i) |  |
|
| (ii) |  |
(a) 1st order |
| (iii) |  |
(b) Zero-order |
| (iv) |  |
Advertisements
Solution
| Column I | Column II | |
| (i) |  |
(a) 1st order |
| (ii) |  |
(b) Zero-order |
| (iii) |  |
(b) Zero order |
| (iv) |  |
(a) 1st order |
Explanation:
For zero order reaction rate equation may be written as [R] = – kt + [R0]
Which denotes a straight line equation similar to y = mx + c
On transmitting (i) `([R] - [R_0])/t = - k`
`k = ([R_0] - [R])/t`
k = Rate
Rate = `k.[t]^0`
Rate `oo [t]^0`
For a first order reaction `(dx)/(dt) oo` [concentration]

∴ Graph between rate and concentration may be drawn as

`k = 2.303/t log ([R]_0)/([R])`
`(kt)/2.303 = log ([R]_0)/([R])`
`(kt)/2.303 = log[R_0] - log[R]`
\[\ce{log[R] = \underset{(Slope)}{\frac{-k}{2.303}} t + \underset{(Intercept)}{log[R]_0}}\]
APPEARS IN
RELATED QUESTIONS
Write molecularity of the following reaction:
2NO(g)+O2(g)→2NO2(g)
For a reaction : 
(i) Write the order and molecularity of this reaction.
(ii) Write the unit of k.
From the rate expression for the following reaction, determine the order of reaction and the dimension of the rate constant.
\[\ce{3NO_{(g)} -> N2O_{(g)}}\] Rate = k[NO]2
A reaction is first order in A and second order in B. Write the differential rate equation.
A reaction is first order in A and second order in B. How is the rate affected when the concentrations of both A and B are doubled?
Rate of reaction for the combustion of propane is equal to:
\[\ce{C3H8_{(g)} + 5O2_{(g)} -> 3CO2_{(g)} + 4H2O_{(g)}}\]
Consider a first order gas phase decomposition reaction given below :
\[\ce{A(g) -> B(g) + C(g)}\]
The initial pressure of the system before decomposition of A was pi. After lapse of time ‘t’, total pressure of the system increased by x units and became ‘pt’ The rate constant k for the reaction is given as ______.
In any unimolecular reaction:
(i) only one reacting species is involved in the rate determining step.
(ii) the order and the molecularity of slowest step are equal to one.
(iii) the molecularity of the reaction is one and order is zero.
(iv) both molecularity and order of the reaction are one.
For a general reaction A → B, plot of concentration of A vs time is given in figure. Answer the following question on the basis of this graph.
(i) What is the order of the reaction?
(ii) What is the slope of the curve?
(iii) What are the units of rate constant?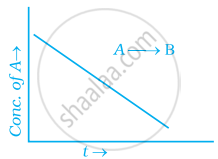
A catalyst in a reaction changes which of the following?
The role of a catalyst is to change
For a reaction A + B → products, the rate law is given by: r = `K[A]^(1/2)`. What is the order of reaction?
The rate constant for the reaction \[\ce{2H2O5 -> 4NO2 + O2}\] is 30 × 10–5 sec–1. if the rate is 204 × 10–5 mol L–1 S–1, then the concentration of N2O5 (in mol–1) is-
If the 0.05 molar solution of m+ is replaced by a 0.0025 molar m+ solution, then the magnitude of the cell potential would be
The number of molecules of the reactants taking part in a single step of the reaction is indicative of ______.
A drop of solution (volume 0.05 ml) contains 3.0 × 10-6 mole of H+. If the rate constant of disappearance of H+ is 1.0 × 107 mole l-1s-1. It would take for H+ in drop to disappear in ______ × 10-9s.
Higher yield of NO in \[\ce{N2(g) + O2 <=> 2NO(g)}\] can be obtained at:
[ΔH of the reaction = +180.7 kJ mol−1]
- higher temperature
- lower temperature
- higher concentration of N2
- higher concentration of O2
Choose the correct answer from the options given below:
