Advertisements
Advertisements
प्रश्न
Match the graph given in Column I with the order of reaction given in Column II. More than one item in Column I may link to the same item of Column II.
| Column I | Column II | |
| (i) |  |
|
| (ii) |  |
(a) 1st order |
| (iii) |  |
(b) Zero-order |
| (iv) |  |
Advertisements
उत्तर
| Column I | Column II | |
| (i) |  |
(a) 1st order |
| (ii) |  |
(b) Zero-order |
| (iii) |  |
(b) Zero order |
| (iv) |  |
(a) 1st order |
Explanation:
For zero order reaction rate equation may be written as [R] = – kt + [R0]
Which denotes a straight line equation similar to y = mx + c
On transmitting (i) `([R] - [R_0])/t = - k`
`k = ([R_0] - [R])/t`
k = Rate
Rate = `k.[t]^0`
Rate `oo [t]^0`
For a first order reaction `(dx)/(dt) oo` [concentration]

∴ Graph between rate and concentration may be drawn as

`k = 2.303/t log ([R]_0)/([R])`
`(kt)/2.303 = log ([R]_0)/([R])`
`(kt)/2.303 = log[R_0] - log[R]`
\[\ce{log[R] = \underset{(Slope)}{\frac{-k}{2.303}} t + \underset{(Intercept)}{log[R]_0}}\]
APPEARS IN
संबंधित प्रश्न
For a reaction A + B ⟶ P, the rate is given by
Rate = k [A] [B]2
How is the rate of reaction affected if the concentration of B is doubled?
For a reaction : 
(i) Write the order and molecularity of this reaction.
(ii) Write the unit of k.
For the hydrolysis of methyl acetate in aqueous solution, the following results were obtained :
| t/s | 0 | 30 | 60 |
| [CH3COOCH3] / mol L–1 | 0.60 | 0.30 | 0.15 |
(i) Show that it follows pseudo first order reaction, as the concentration of water remains constant.
(ii) Calculate the average rate of reaction between the time interval 30 to 60 seconds.
(Given log 2 = 0.3010, log 4 = 0.6021)
The conversion of molecules X to Y follows second order kinetics. If concentration of X is increased to three times how will it affect the rate of formation of Y?
From the rate expression for the following reaction, determine the order of reaction and the dimension of the rate constant.
\[\ce{H2O2_{( aq)} + 3I^-_{( aq)} + 2H^+ -> 2H2O_{(l)} + I^-_3}\] Rate = k[H2O2][I−]
Mention the factors that affect the rate of a chemical reaction.
How does calcination differ from roasting?
What is the order of a reaction which has a rate expression; Rate = `"k"["A"]^(3/2)["B"]^1`?
In a reaction if the concentration of reactant A is tripled, the rate of reaction becomes twenty seven times. What is the order of the reaction?
For a general reaction A → B, plot of concentration of A vs time is given in figure. Answer the following question on the basis of this graph.
(i) What is the order of the reaction?
(ii) What is the slope of the curve?
(iii) What are the units of rate constant?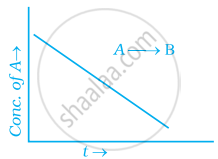
The role of a catalyst is to change
The rate constant for the reaction \[\ce{2H2O5 -> 4NO2 + O2}\] is 30 × 10–5 sec–1. if the rate is 204 × 10–5 mol L–1 S–1, then the concentration of N2O5 (in mol–1) is-
If the 0.05 molar solution of m+ is replaced by a 0.0025 molar m+ solution, then the magnitude of the cell potential would be
For a reaction 1/2 A ⇒ 2B, rate of disappearance of A is related 't o the appearance of B by the expression:
The number of molecules of the reactants taking part in a single step of the reaction is indicative of ______.
For a chemical reaction starting with some initial concentration of reactant At as a function of time (t) is given by the equation,
`1/("A"_"t"^4) = 2 + 1.5 xx 10^-3` t
The rate of disappearance of [A] is ____ × 10-2 M/sec when [A] = 2 M.
[Given: [At] in M and t in sec.]
[Express your answer in terms of 10-2 M /s]
[Round off your answer if required]
Assertion (A): Order of reaction is applicable to elementary as well as complex reactions.
Reason (R): For a complex reaction, molecularity has no meaning.
Which of the following statement is true?
A reaction is second order with respect to a reactant. How is the rate of reaction affected if the concentration of the reactant is reduced to half?
