Advertisements
Advertisements
प्रश्न
Write the mechanism of the following reaction:

Advertisements
उत्तर
The mechanism of the given reaction is as follows:
(i) Protonation of an alcohol molecule
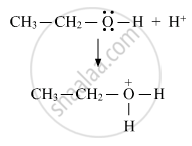
(ii) Attack of other alcohol molecule on the protonated molecule
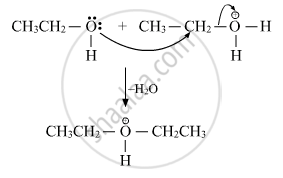
(iii) Deprotonation
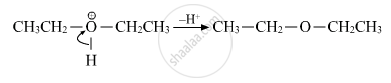
APPEARS IN
संबंधित प्रश्न
Give reasons for the following:
o-nitrophenol is more acidic than o-methoxyphenol.
Name the reagent used in the following reaction:
Benzyl alcohol to benzoic acid.
Lucas reagent is ____________.
By which of the following methods alcohol can be prepared in excellent yield?
Which one of the following on oxidation gives a ketone?
Cyclohexene is best prepared from cyclohexanol by which of the following:
Suggest a reagent for the following conversion.

In Kolbe’s reaction, instead of phenol, phenoxide ion is treated with carbon dioxide. Why?
The correct geometry around oxygen in CH3OCH3 is
Write the mechanism of acid dehydration of ethanol to yield ethene.
