Advertisements
Advertisements
प्रश्न
Alcohols are comparatively more soluble in water than hydrocarbons of comparable molecular masses. Explain this fact.
Advertisements
उत्तर
Alcohols are more soluble in water compared to hydrocarbons with equivalent molecular weight because alcohol molecules form hydrogen bonds with water and can also break the existing hydrogen bonds between water molecules, while hydrocarbons are unable to do this.
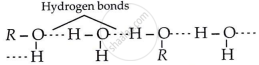
APPEARS IN
संबंधित प्रश्न
Explain why propanol has higher boiling point than that of the hydrocarbon, butane?
Give reason for the higher boiling point of ethanol in comparison to methoxymethane.
`CH_3-CH_2-Br"Alcoholic KOH"/""> B " HBR"/"">C"Na/Eather"/"">D`, the Compound D is
(A) ethane
(B) propane
(C) n-butane
(D) n-pentane
Arrange the following compounds in the increasing order of their acid strength:
p-cresol, p-nitrophenol, phenol
Identify the chiral molecule in the following pair :

Write the structures of A, B and C in the following reactions :
\[ C_6 H_5 {NO}_2 \to^{Sn/HCI} A \to^{{NaNO}_2 /HCI}_{273 K} B \to^{H_2 O}_∆ C\]
Account for the following:
CH3CHO is more reactive than CH3COCH3 towards reaction with HCN.
Write a chemical reaction to show that the open structure of D-glucose contains the following :
Five alcohol groups
Isopropyl alcohol is obtained by reacting which of the following alkenes with concentrated H2SO4 followed by boiling with H2O?
Which of the following has lowest boiling point?
Which statement is not correct about alcohol?
Explain why alcohols and ethers of comparable molecular mass have different boiling points?
Assertion: Boiling points of alcohols and ethers are high.
Reason: They can form intermolecular hydrogen-bonding.
Assertion (A): Alcohols react both as nucleophiles and electrophiles.
Reason (R): The bond between C–O is broken when alcohols react as nucleophiles.
Select the most appropriate answer from the options given below:
How are the following conversion carried out?
Methyl magnesium bromide →2-Methylpropan-2-ol.
Convert the following:
acetaldehyde to isopropyl alcohol.
How are the following conversions carried out?
\[\ce{Methyl magnesium bromide -> 2-Methylpropan-2-ol}\]
