Advertisements
Advertisements
प्रश्न
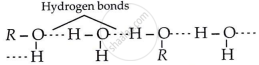
Alcohols are comparatively more soluble in water than hydrocarbons of comparable molecular masses. Explain this fact.
Advertisements
उत्तर
Alcohols are more soluble in water compared to hydrocarbons with equivalent molecular weight because alcohol molecules form hydrogen bonds with water and can also break the existing hydrogen bonds between water molecules, while hydrocarbons are unable to do this.
APPEARS IN
संबंधित प्रश्न
Give reasons for the following : Boiling point of ethanol is higher in comparison to methoxymethane.
`CH_3-CH_2-Br"Alcoholic KOH"/""> B " HBR"/"">C"Na/Eather"/"">D`, the Compound D is
(A) ethane
(B) propane
(C) n-butane
(D) n-pentane
Give simple chemical tests to distinguish between the following pairs of compounds :
Ethanol and phenol
Account for the following:
CH3CHO is more reactive than CH3COCH3 towards reaction with HCN.
Alcohols have high boiling points because of ____________.
Isopropyl alcohol is obtained by reacting which of the following alkenes with concentrated H2SO4 followed by boiling with H2O?
The correct order of boiling points for primary (1°), secondary (2°) and tertiary alcohol (3°) is:
Which statement is not correct about alcohol?
Which one of the following alcohols is least soluble in water?
Explain why alcohols and ethers of comparable molecular mass have different boiling points?
A solution of phenol in chloroform when treated with aqueous NaOH gives compound P as a major product. The mass percentage of carbon in P is ______. (to the nearest integer) (Atomic mass: C = 12; H = 1; O = 16)
Write a note on Kolbe's reaction
How are the following conversions carried out?
Methyl magnesium bromide → 2 -Methylpropan-2-ol.
Convert the following:
acetaldehyde to isopropyl alcohol.
How are the following conversions carried out?
\[\ce{Methyl magnesium bromide ->2-Methylpropan-2-ol}\]
How are the following conversions carried out?
\[\ce{Methyl magnesium bromide -> 2-Methylpropan-2-ol}\]
How is the following conversion carried out?
\[\ce{Methyl magnesium bromide ->2-Methylpropan-2-ol}\]
