Advertisements
Advertisements
प्रश्न
Explain why propanol has higher boiling point than that of the hydrocarbon, butane?
स्पष्ट करा
Advertisements
उत्तर
The boiling point of a compound is dependent upon the strength of intermolecular interactions. The greater the intermolecular attraction, the higher the boiling point.
In butane, the molecules engage with one another via weak van der Waals forces. These weak forces can be easily overcome by providing a small amount of thermal energy. Thus, they have a low boiling point.
The molecules in propanol are held together by strong hydrogen bonds. When molecules are attracted to each other, it’s harder to break those bonds. This means more heat is needed, which raises the boiling point.
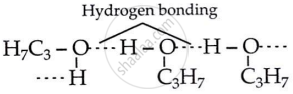
shaalaa.com
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
