Advertisements
Advertisements
प्रश्न
Identify allylic alcohols in the following examples.
विकल्प
\[\begin{array}{cc}
\ce{CH3}\phantom{.}\\
|\phantom{.....}\\
\ce{CH3 - C - CH2OH}\\
|\phantom{.....}\\
\ce{CH3}\phantom{.}
\end{array}\]H2C = CH – CH2OH
CH3 – CH2 – CH2 – OH
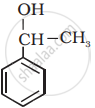
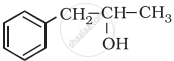

MCQ
Advertisements
उत्तर
H2C = CH – CH2OH and 
shaalaa.com
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
अध्याय 7: Alcohols, Phenols and Ethers - Intext Questions [पृष्ठ १९५]
