Advertisements
Advertisements
प्रश्न
Write the mechanism of the reaction of HI with methoxymethane.
Advertisements
उत्तर
The reaction between methoxymethane and HI is:
\[\ce{CH3 - O - CH3 + HI -> CH3I + CH3OH}\]
Generation of oxonium ion (Step 1): Protonation of the ether molecule.
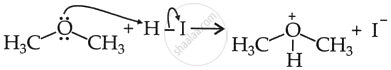
Nucleophilic attack on oxonium ion (Step 2):
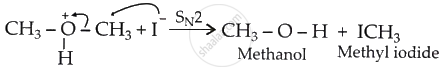
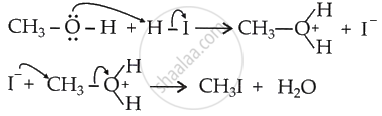
If excess HI is used, the methanol formed above reacts with HI to form methyl iodide.
संबंधित प्रश्न
Write the main product(s) in each of the following reactions:

Predict the product of the following reaction:
\[\ce{CH3 - CH2 - CH2 - O - CH3 + HBr ->}\]
Predict the product of the following reaction:
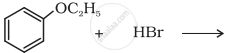
Write the equation of the reaction of hydrogen iodide with 1-propoxypropane.
Write the equation of the reaction of hydrogen iodide with benzyl ethyl ether.
Explain the fact that in aryl alkyl ethers
- the alkoxy group activates the benzene ring towards electrophilic substitution and
- it directs the incoming substituents to ortho and para positions in the benzene ring.
Write the formula of reagents used in the following reactions :
Bromination of phenol to 2,4,6-tribromophenol
Write the formula of reagents used in the following reactions :
Hydroboration of propene and then oxidation to propanol.
Write the structures of the main products in the following reactions :

The ether
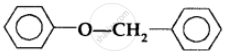
when treated with HI produces:
The major organic product in the reaction is \[\mathrm{CH}_{3}-\mathrm{O}-\mathrm{CH}(\mathrm{CH}_{3})_{2}+\mathrm{HI}\to\mathrm{products}\]:
Consider the following reaction
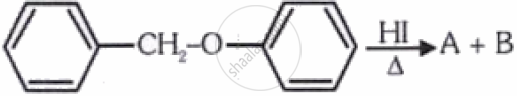
Identify products A and B:-
