Advertisements
Advertisements
Question
Write the mechanism of the reaction of HI with methoxymethane.
Advertisements
Solution
Methoxymethane and equimolar amounts of HI form a mixture of methyl alcohol and methyl iodide. The mechanism of the reaction is as follows –
Step 1: Generation of oxonium ion:
\[\begin{array}{cc}
\phantom{..............................}\ce{H}\\
\phantom{..............................}|\\
\ce{\underset{Methoxymethane}{CH3 - \overset{\bullet\bullet}{\underset{\bullet\bullet}{O}} - CH3 + H - I} ->[Protonation][rapid] \underset{Dimethyl oxonium ion}{CH3 - \overset{+}{\underset{\bullet\bullet}{O}} - CH3 + I^-}}
\end{array}\]
Step 2: Nucleophilic attack on oxonium ion:
\[\begin{array}{cc}\\
\ce{H}\phantom{......................}\\
|\phantom{......................}\\
\ce{I^- + CH3 - O^+ - CH3 ->[S_{N}2][dim] \underset{Methyl iodide}{CH3 - I} + \underset{Methanol}{CH3OH}}
\end{array}\]
If excess of HI is used then methanol formed in the second step gets converted into methyl iodide by the following mechanism:
\[\begin{array}{cc}
\phantom{............................}\ce{H}\\
\phantom{............................}|\\
\ce{CH3 - \overset{\bullet\bullet}{\underset{\bullet\bullet}{O}} - H + H - I ->[Protonation][dim] \underset{Protonated Methanol}{CH3 - \underset{\bullet\bullet\phantom{...}}{O^+} - H + I^-}}
\end{array}\]
\[\begin{array}{cc}
\ce{H}\phantom{.................}\\
|\phantom{.................}\\
\ce{I^- + CH3 - \underset{\bullet\bullet\phantom{...}}{O^+} - H ->[S_{N}2][dim] \underset{Methyl iodide}{CH3 - I} + H2O}
\end{array}\]
RELATED QUESTIONS
Write the main product(s) in each of the following reactions:

Predict the product of the following reaction:
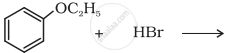
Predict the product of the following reaction:
\[\ce{(CH3)3C - OC2H5 ->[HI]}\]
Write the equation of the reaction of hydrogen iodide with 1-propoxypropane.
Write the equation of the reaction of hydrogen iodide with methoxybenzene.
Write the equation of the reaction of hydrogen iodide with benzyl ethyl ether.
Explain the fact that in aryl alkyl ethers
- the alkoxy group activates the benzene ring towards electrophilic substitution and
- it directs the incoming substituents to ortho and para positions in the benzene ring.
Write the product(s) in the following reaction
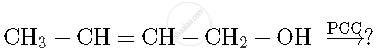
Write the formula of reagents used in the following reactions :
Bromination of phenol to 2,4,6-tribromophenol
Write the formula of reagents used in the following reactions :
Hydroboration of propene and then oxidation to propanol.
Write the structures of the main products in the following reactions :

Maximum number of H-bonds that can be formed by a water molecule is
Indicate the σ and π bond in the following molecule:
HCONHCH3
The major organic product in the reaction is \[\mathrm{CH}_{3}-\mathrm{O}-\mathrm{CH}(\mathrm{CH}_{3})_{2}+\mathrm{HI}\to\mathrm{products}\]:
