Advertisements
Advertisements
प्रश्न
You are given benzene, conc. H2SO4 and NaOH. Write the equations for the preparation of phenol using these reagents.
Advertisements
उत्तर १

उत्तर २
\[\ce{\underset{Benzene}{C6H6} ->[conc.H2SO4/ \Delta][sulfonation] \underset{Benzene sulphonic acid}{C6H5SO3H} ->[NaOH, melting][-Na2SO3] \underset{Sodium phenoxide}{C6H5ONa} ->[Dil. HCl][-NaCl] \underset{Phenol}{C6H5OH}}\]
APPEARS IN
संबंधित प्रश्न
Give the equations of reactions for the preparation of phenol from cumene.
Write chemical reaction for the preparation of phenol from chlorobenzene.
Write the structures of main products when benzene diazonium chloride (C6 H5 N+2CI−)C6 H5 N2+CI- reacts with the : HBF4/∆
Write the structures of main products when benzene diazonium chloride (C6 H5 N+2CI−)C6 H5 N2+CI- reacts with the :Cu/HBr
Why phenol undergoes electrophilic substitution more easily than benzene?
Sodium salt of benzene sulphonic acid on fusion with caustic soda gives ____________.
Which of the following will not form phenol or phenoxide?
Which of the following reactions will yield phenol?
| (i) |  |
| (ii) | 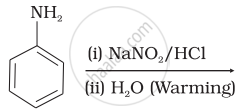 |
| (iii) | 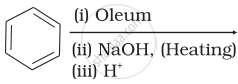 |
| (iv) |  |
Name the starting material used in the industrial preparation of phenol.
Benzoic acid reacts with cone. HNO3 and HiSO4 to give:
Name the catalyst used in the commercial method of preparation of phenol.
In the reaction, alcohol + diazomethane → ether + N2; the ether cannot be ______.

Consider the above reactions, the product A and product B respectively are:
'A' and 'B' in the following reactions are:

