Advertisements
Advertisements
प्रश्न
What happens when Phenol is oxidised with Na2Cr2O7/H+?
What happens when Phenol is oxidised with Na2Cr2O7/H+?
Write the chemical equation in support of your answer.
Advertisements
उत्तर
When phenol is treated with chromic acid (Na2Cr2O7 + H+), it generates benzoquinone, a conjugated diketone.

APPEARS IN
संबंधित प्रश्न
How do you convert the following:
Aniline to phenol
Give the equations of reactions for the preparation of phenol from cumene.
Write the structures of main products when benzene diazonium chloride (C6 H5 N+2CI−)C6 H5 N2+CI- reacts with the :Cu/HBr
Which of the following reactions will yield phenol?
| (i) |  |
| (ii) | 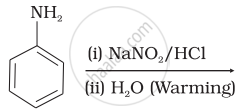 |
| (iii) | 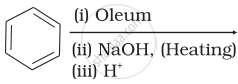 |
| (iv) |  |
Name the starting material used in the industrial preparation of phenol.
What happens when reactions:
N-ethylethanamine reacts with benzenesulphonyl chloride.
Benzoic acid reacts with cone. HNO3 and HiSO4 to give:
Aspirin is also known as
In the reaction, alcohol + diazomethane → ether + N2; the ether cannot be ______.

Consider the above reactions, the product A and product B respectively are:
