Advertisements
Advertisements
प्रश्न
Which of the following are benzylic alcohols?
(i) \[\ce{C6H5 - CH2 - CH2OH}\]
(ii) \[\ce{C6H5 - CH2OH}\]
(iii) \[\begin{array}{cc}
\ce{C6H5 - CH - OH}\\
\phantom{}|\phantom{.}\\
\phantom{..}\ce{CH3}\phantom{}
\end{array}\]
(iv) \[\begin{array}{cc}
\ce{C6H5 - CH2 - CH - OH}\\
\phantom{.......}|\phantom{}\\
\phantom{.........}\ce{CH3}\phantom{}
\end{array}\]
Advertisements
उत्तर
(ii) \[\ce{C6H5 - CH2OH}\]
(iii) \[\begin{array}{cc}
\ce{C6H5 - CH - OH}\\
\phantom{}|\phantom{.}\\
\phantom{..}\ce{CH3}\phantom{}
\end{array}\]
Explanation:
In these alcohols, the –OH group is attached to a sp3- hybridised carbon atom next to an aromatic ring. For example,
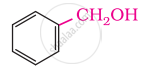
Primary
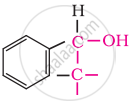
Secondary
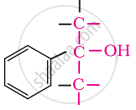
Tertiary
Allylic and benezlic alcohols may be primary, secondary or tertiary.
APPEARS IN
संबंधित प्रश्न
Write the final product(s) in each of the following reactions:

Write the main product(s) in each of the following reactions:

Name the reagent used in the following reaction:
Bromination of phenol to 2, 4, 6-tribromophenol.
Write the reaction involved in the following:
Friedal-Crafts Alkylation of Phenol
On distilling phenol with Zn dust, one gets:
The electrophile involved in Reimer-Tiemann reaction of phenol with CHCl3 in presence of NaOH:

Phenol does not undergo nucleophilic substitution reaction easily due to ______.
Which of the following compounds is aromatic alcohol?
| (A) |  |
| (B) | 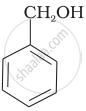 |
| (C) |  |
| (D) |  |
Which of the following are used to convert \[\ce{RCHO}\] into \[\ce{RCH2OH}\]?
(i) \[\ce{H2/Pd}\]
(ii) \[\ce{LiAlH4}\]
(iii) \[\ce{NaBH4}\]
(iv) Reaction with \[\ce{RMgX}\] followed by hydrolysis
Convert the following:
Phenol to N-phenylethanamide.
Attacking species in nitration of benzene in presence of fuming HNO3 is
Write the equations for the following reaction:
Phenol is treated with chloroform in the presence of NaOH
Why ortho-nitrophenol is steam volatile while para-nitrophenol is not?
Write the chemical equation involved in the following reactions:
Acetylation of salicylic add
For the pair phenol and cyclohexanol, answer the following:
Give one chemical test to distinguish between the two.
How can phenol be prepared from anisole? Give reaction.
Convert the following by giving a chemical equation:
Phenol to salicylaldehyde.
