Advertisements
Advertisements
प्रश्न
Write the name of the reaction, structure and IUPAC name of the product formed when:
Phenol reacts with CHCl3 in the presence of NaOH followed by hydrolysis.
Advertisements
उत्तर
Reimer Tiemann

APPEARS IN
संबंधित प्रश्न
Give simple chemical tests to distinguish between the following pairs of compounds: Benzoic acid and Phenol
While separating a mixture of ortho and para nitrophenols by steam distillation, name the isomer which will be steam volatile. Give reason.
When phenol is heated with CHCl3 and alcoholic KOH when salicylaldehyde is produced. This reaction is known as ____________.
In the reaction of phenol with CHCl3 and aqueous NaOH at 343 K, the electrophile attacking the ring is:
\[\ce{C2H5OH + SOCl2 ->[Pyridine] C2H5Cl + SO2 + HCl}\]
The above reaction is known as:
Which of the following reactions is used to prepare salicylaldehyde?
Which of the following compounds is aromatic alcohol?
| (A) |  |
| (B) | 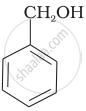 |
| (C) |  |
| (D) |  |
Out of o-nitrophenol and p-nitrophenol, which is more volatile? Explain.
Nitration is an example of aromatic electrophilic substitution and its rate depends upon the group already present in the benzene ring. Out of benzene and phenol, which one is more easily nitrated and why?
Convert the following by giving a chemical equation:
Phenol to salicylaldehyde.
