Advertisements
Advertisements
प्रश्न
Account for the following.
Ethylamine is soluble in water whereas aniline is not.
State reasons for the following:
Ethylamine is soluble in water whereas aniline is insoluble in water.
Advertisements
उत्तर १
Ethylamine, when added to water, forms intermolecular H-bonds with water and therefore it is soluble in water. But aniline does not form an H-bond with water to a very large extent due to the presence of a large hydrophobic –C6H5 group. Hence, aniline is insoluble in water.
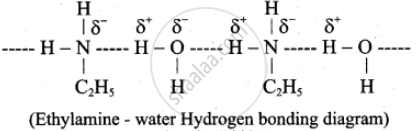
उत्तर २
Ethylamine (C2H5NH2) exhibits solubility in water because the –NH group effectively forms hydrogen bonds with water molecules, due to the increased electron density on the nitrogen atom resulting from the +I inductive effect of the –C2H5 group. Also, the hydrocarbon part, –C2H5, is small and contributes much to hydrophobicity.
In Aniline (C6H5NH2), the –NH2 group is involved in resonance with the directly attached benzene ring, hence limiting its ability to form effective hydrogen bonds with water. Secondly, the benzene ring is substantial and generally hydrophobic. Hence, the net result is the insolubility of aniline in water.
संबंधित प्रश्न
Write the number of moles of ethanoyl chloride required for complete acylation of N, N-dimethylaniline.
What is the action of nitrous acid on the following compounds?
Isopropyl amine
The order of basic strength for methyl substituted amines in aqueous solution is ____________.
Write a short note on the following.
Ammonolysis
Write a short note on the following:
Gabriel phthalimide synthesis
Write a short note on the following.
Carbylamine reaction
How will you prepare propan-1-amine from propanamide?
The reaction of NHO2 with 'A' gives quartering ammonium salt. A is which of the following?
Assertion A: Aniline on nitration yields ortho, meta and para nitro derivatives of aniline.
Reason R: Nitrating mixture is a storng acidic mixture.
In the light of the above statements, choose the correct answer from the options given below:
Account for the following:
Aniline does not undergo Friedel-Crafts reaction.
