Advertisements
Advertisements
प्रश्न
Classify the following amine as primary, secondary or tertiary:
(C2H5)2CHNH2
विकल्प
Primary
Secondary
Tertiary
Advertisements
उत्तर
Primary
APPEARS IN
संबंधित प्रश्न
In the following

The compound ‘B’ is _______.
(A) Propan–1–amine
(B) Propan–2–amine
(C) Isopropylamine
(D) Dimethylamine
How are amines classified?
\[\ce{Aniline + benzoylchloride ->[NaOH] C6H5 - NH - COC6H5}\] this reaction is known as ____________.
The product formed by the reaction an aldehyde with a primary amine ____________.
When aniline reacts with acetic anhydride the product formed is ____________.
Write a short note on the following.
Schotten-Baumann reaction
Write a short note on the following.
Mustard oil reaction
Account for the following.
Gabriel phthalimide synthesis is preferred for synthesising primary amines.
How will you prepare propan-1-amine from butane nitrile?
Identify A, B and C.
\[\ce{CH3 - NO2 ->[LiAlH4] A ->[2CH3CH2Br] B ->[H2SO4] C}\]
(C2H5)2CHNH2. The following amine can be classified as:
Arrange the following in the increasing order of their basic strength:
C2H5NH2, C6H5NH2, (C2H5)2NH
\[\begin{array}{cc}
\ce{O}\phantom{.........}\\
||\phantom{.........}\\
\ce{H - \underset{(A)}{C} - NH - CH3}
\end{array}\]
and
\[\begin{array}{cc}
\ce{O}\\
||\\
\ce{CH3 - \underset{(B)}{C} - NH2}
\end{array}\]
are which type of isomers?
Classify the following amine as primary, secondary or tertiary:
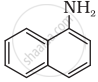
Arrange the increasing order of solubility in water.
\[\ce{C2H5Cl, C2H5NH2, C2H5OH}\]
Write short note on the following:
Ammonolysis
Name the distinguishing test for differentiating 1° amine from 2° and 3° amine.
Define Amines.
Write a short note on the following.
Ammonolysis
Write short note on the following:
Ammonolysis
