Advertisements
Advertisements
प्रश्न
Classify the following amine as primary, secondary or tertiary:
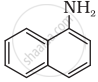
विकल्प
Primary
Secondary
Tertiary
Advertisements
उत्तर
Primary
APPEARS IN
संबंधित प्रश्न
Classify the following amine as primary, secondary or tertiary:
(C2H5)2NH
Isobutylamine is an example of ______.
Choose the most correct option.
Which type of amine does produce N2 when treated with HNO2?
How are amines classified?
Write the number of moles of ethanoyl chloride required for complete acylation of N, N-dimethylaniline.
A tertiary amine is an organic compound ____________.
Assertion: Acetamide on reaction with KOH and bromine gives acetic acid.
Reason: Bromine catalyses hydrolysis of acetamide.
Which one of the following is most basic?
IUPAC name for the amine is:
\[\begin{array}{cc}
\phantom{.}\ce{CH3}\\
|\phantom{..}\\
\ce{CH3 - N - C - CH2 - CH3}\\
\phantom{.}|\phantom{.....}|\phantom{........}\\
\phantom{}\ce{CH3}\phantom{..}\ce{C2H5}\phantom{....}
\end{array}\]
Write a short note on the following.
Schotten-Baumann reaction
Arrange the following:
In increasing order of solubility in water:
C6H5NH2, (C2H5)2NH, C2H5NH2
Identify A, B and C.
\[\ce{CH3 - NO2 ->[LiAlH4] A ->[2CH3CH2Br] B ->[H2SO4] C}\]
Arrange the following in the increasing order of their basic strength:
C2H5NH2, C6H5NH2, (C2H5)2NH
The amine formed from an amide by mean of bromine and alkali has how many number of carbon atoms?
Which among the following is the strongest Bronsted base?
Write short note on the following.
Ammonolysis
Write short note on the following:
Ammonolysis
